subtidal rocky areas discussed in
Chapter 5. Most of the dominant organisms of intertidal rocks are solitary or
clonal animals, whereas subtidal rocks seem dominated by colonial encrusting
animals.
Zonation
One of the most striking features of any rocky shore anywhere
in the world at low tide is the prominent horizontal banding or zonation of the organisms. Each zone or
band is set off from those adjacent by differences in color, morphology of the
major organism, or some combination of color and morphology. These horizontal
bands or zones succeed each other vertically as one progresses
up from the level of the lowest low tides to true terrestrial conditions
(Figures 6.7, 6.8). This zonation on intertidal rocky shores is similar to the
zonation pattern one observes with increasing elevation on a mountain, where
the different horizontal zones of trees and shrubs succeed each other
vertically until, if one progresses far enough, permanent snow cover is
reached. The major difference between these two areas is the scale. Mountain
zones are perhaps kilometers in extent as opposed to intertidal zones
extending a few meters vertically.
Rocky intertidal zones vary in vertical extent, depending on
the slope of the rocky surface, the tidal range, and the exposure to wave
action. Where there is a gradual slope to the rock, individual zones may be
broad. Under similar tidal and exposure conditions on a vertical face, the
same zones would be narrow. In the same manner, exposed areas have broader
zones than protected shores, and shores with greater tidal ranges have broader
vertical zones (Figures 6.4, 6.8).
Of course, these striking bands may be interrupted or altered
in various places wherever the rock substrate shows changes in slope, composition,
or irregularities that change its exposure or position relative to the
prevailing water movement.
The fact that these prominent zones can be observed on nearly
all rocky shores throughout the world under many different tidal regimes led
Stephenson and Stephenson (1949) to propose, after some 30 years of study, a
"universal" scheme of zonation for rocky shores (Figure 6.9). This
universal scheme was really a framework using common terms that would allow
comparison of diverse areas. It established zones based on the distributional
limits of certain common groups of organisms and not on tides. It reflects the
knowledge of the Stephensons, and other intertidal ecologists, that
distribution patterns of the organisms and zones vary not only with tides, but
with slopes and exposure. Therefore, under similar tidal conditions there could
be different bandwidths due to different exposures or slopes of rocks. It was
this universal scheme that established a standard format for describing shore
zonation, replacing a bewildering host of schemes and names established by
earlier biologists.
The Stephensons' scheme has three main divisions of the
intertidal area. The uppermost is termed the supralittoral fringe. Its lower limit is the upper limit of
barn4eles, and it extends to the upper limit of snails of the genus Littorina (periwinkles). The dominant
organisms are the littorine snails and black encrusting lichens (Verrucaria type). The extreme high
water of spring tides reaches part of this zone, but most of its water comes
from wave splash. Above this zone is the terrestrial or supralittoral zone.
The middle part of the intertidal
is termed the midlittoral zone and
is the broadest in extent. Its upper limit coincides with the upper limit of barnacles,
while its lower limit is the point where
zone is often subdivided and contains a host of different
organisms. Perhaps the only universally present dominant group
are the barnacles.
The lowest zone of this scheme is the infralittoral fringe, which extends
from the lowest low tide up to the upper limit of the large kelps. This is an
extremely rich zone composed of organisms that can tolerate only limited
exposure to air. It is really an intertidal extension of the infralittoral zone (Stephensons' term)
or what we know as the sublittoral area.
Although Stephensons' scheme did set forth a
means for describing zonation on rocky shores, it does not offer an explanation
of why the zonation occurs. It is this explanation of zonal patterns that
intrigues many marine biologists.
Causes of Zonation
Whereas
it is fairly easy to recognize and measure the extent of the zones on a rocky
shore, it is more difficult to find suitable explanations for why organisms
are distributed in these zones. Physical and biological factors can be
considered to explain the phenomenon. We shall take up each in turn.
Upper-limit
barnacles
Equinoctial
high-water spring tides
,aria
is
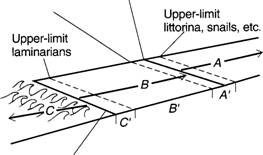 A = Supralittoral (spray)
zone A' = Supralittoral fringe
A = Supralittoral (spray)
zone A' = Supralittoral fringe
B = Littoral zone
B' = Midlittoral
C = Infralittoral zone C
= Infralittoral fringe
FIGURE 6.9 Stephensons'
universal scheme of zonation for rocky shores. (From R. L.
Smith, Ecology and field biology, 3d ed., Figure 8-4. Copyright © 1980
by Robert
Leo Smith. Reprinted
by permission of Harper & Row Publishers, Inc.)
large kelps (Laminaria, etc.)
reach their uppermost distribution. This
nalus is
aris
Equinoctial low-water
spring tides
ienia
PHYSICAL FACTORS The most obvious explanation for the occurrence of the zones
is that they are a result of the tidal action on the shore and reflect the
different tolerances of the organisms to increasing exposure to the air and
the resultant desiccation and temperature extremes. One difficulty with this
explanation is that the rise and fall of a tide tend to follow a smooth curve
with, no obvious sharp breaks corresponding to the often sharp
boundaries observed in the intertidal zones. If, however, one observes a whole
series of such tidal curves, such a series of breaks does become apparent. For
example, Figure 6.10a gives a typical tidal curve for a mixed tide area on the
the early explanations for the
zonation patterns described previously. In this explanation, it is important
to remember that it is not the tide alone that causes the limit but the fact
that, at these critical points, the organisms are subjected to greatly increased
time in the air; hence, they experienced greater temperature fluctuations and
desiccation.
The critical tide hypothesis has been tested in various
places by several scientists since its original promulgation by Doty in 1946.
In general, it is difficult to find good correlations, particularly at the
lower tide levels. This lack of good correlation can be partly attributed to
the diverse topography of the different shores and to the variation in
exposure. Thus, a species may be able to exist above the critical tide level if
the rocky shore is exposed to persistent violent wave action, which would
throw water up higher and decrease desiccation. Similarly, caves, overhangs,
and crevices remain moist when exposed areas are dried out; thus, organisms
are able to persist above the critical tide level. The effects of differences
in topography and exposure suggest, however, that the ultimate upper limits are
set by certain physical factors: desiccation and temperature.
We have previously noted that desiccation is a serious
problem. Several natural observations, as well as field experiments, have
suggested that desiccation can set the upper limits to organisms and zones. In
the
rocks that have a north-facing
slope often have the same organism occurring higher than do adjacent
south-facing slopes. Since such rocks experience no difference in wave exposure
or tides, the only reason for the height difference is that north faces dry out
more slowly than south faces. Similarly, field experiments by Frank (1965) and
others in which water was slowly dripped down slopes have resulted in the
organisms extending higher up (Figure 6.11). In other experiments, organisms
were transplanted above their normal positions. Notable in this respect are
barnacles, which cannot move. When transplanted by Foster (1971) above their
normal tidal height, they died. Younger barnacles died more quickly than older
ones. Again, desiccation can be suspected.
Along with desiccation, and often acting in concert with it,
is temperature. As noted previously, aerial temperatures have ranges that may
exceed the lethal limits; hence, intertidal organisms may die from either
freezing or "cooking." Upper limits of zones in part may be
attributed to the tolerable temperature limits of the intertidal organisms. In
addition, high temperature promotes desiccation, and the synergistic effect of
these factors may be even more devastating than each acting alone. The lack of
abundant organisms on rocky intertidal shores of the tropics may be at least
partly the result of desiccation combined with heat and ultraviolet light
stress.
Finally, sunlight may act adversely to limit organisms on the
shore. Sunlight includes wavelengths in the ultraviolet (UV) region that can
harm living tissue. Water absorbs these wavelengths and protects most marine
animals. However, intertidal animals have direct exposure to such rays at low
tide. The higher an organism is in the intertidal, the greater the exposure to
these rays. At present, there is little information concerning whether UV
radiation controls distribution of organisms, but the recent finding of
UV-absorbing compounds in shallow-water organisms suggests its importance.
Light also has been suggested as a regulator of the distribution of intertidal
algae. This has to do primarily with the spectral quality of the light. As
noted in Chapter 1, different wavelengths of light are absorbed differentially
by water. In the case of intertidal algae, those that need light of longer
wavelength (reds) that are absorbed most quickly by water would tend to be
found higher in the intertidal. When submerged, they would not be too deep for
the penetration of red light (about 2 m). Since the main intertidal algae
belong to three different groups-reds, browns, and greens-each of which has a
slightly different absorption spectrum, it might be thought that they would be
arranged along a depth gradient. In such a gradient, the green algae would be
expected to be the highest, because they absorb mainly red light; the brown
next; and the red algae deepest, because they absorb mainly the deep,
penetrating green light. This, however, is not the case. The intertidal algae
are a mixture of all types at most levels. This is likely due to the
interaction of other factors and the physiology of the algae. It points out
again the fallacy of attempting to explain patterns of species distribution
with only single factors when a multitude are acting
at all times.
Although there is a large body of evidence that physical
factors are the strongest determinants of the upper limits of organisms, there
may be alternative explanations, many of which have not been investigated.
Underwood and Denley (1984) point out that other processes are at work in the
high intertidal that may be confused with physical effects. For example, some
animals in the high intertidal, particularly sessile filter feeders, may be
killed by starvation during long periods of calm weather because they are not
covered with water long enough to feed. Similarly, grazing animals may be
absent, not because they cannot tolerate the physical factors, but because
their algal food is absent, perhaps killed by severe physical factors. Finally,
in the case of larvae settling out of the plankton, they may choose not to
settle in the area, in which case the upper limit is set by processes affecting
larval settlement. In one instance reported by Underwood (1980), the upper
limit of several algal species in
BIOLOGICAL FACTORS Although the early work in the intertidal focused on the
importance of physical factors in setting the zonal patterns, more recent work
has begun to clearly establish the great importance of a number of biological
factors in setting various observed distribution patterns. In general, these
biological factors are more complex, often subtle, and closely linked to other
factors. This is probably why we have only recently begun to understand how
they act. The major biological factors are competition,
predation, grazing (herbivory), and larval
settlement. We shall discuss them in order.
Competition for a certain resource does not occur if the
resource is so plentiful that adequate supplies of it are available for all
species or individuals. In the rocky intertidal zone, only one resource is
commonly in limited supply and that is space. This is perhaps the most
restricted area in the marine environment; at the same time, it is densely
populated, at least in the temperate zone. As a result, there is an intense
competition for space that has resulted in observed zonal patterns.
On the intertidal shores of
Chthamalus stellatus living in the highest zone and the larger Balanus balanoides occupying the
major portion of the
midintertidal. Studies done by Connell (1961) showed that Chthamalus larvae settled out throughout the zones occupied by both
barnacles but survived to adulthood only in upper
zones.
The reason for the disappearance in the midlittoral region was due to
competition from Balanus balanoides, which
either overgrew, uplifted, or crushed the young Chthamalus. Balanus was prevented from
completely eliminating Chthamalus, because
unlike Cbthamalus, Balanus did not
have the tolerance to drying and high temperatures that prevailed at the
higher tidal levels; hence, it could not survive there. Here, then, is a case
where the zonation is at least partially a function of biological competition
(Figure 6.12).
Intertidal algae, particularly those on temperate shores, also
often show abrupt limits to their upper and lower distribution. Earlier, these
limits were ascribed to critical tide levels, but it has also been suggested
that this could be due to competition for space or access to light. In two
experimental studies done on the Pacific coast of
Competition among the
mussel Mytilus californianus and
several species of barnacles on the Pacific coast of
ese, in turn, displace any rapidly growing algal
species. The barnacles persist
only until the mussels enter. The mussels outcompete and destroy the barnacles
by settling on top of them and smothering them. Since nothing appears to be
large enough to settle and smother the M.
californianus, they remain in control of the intertidal space. Given this
competitive edge, it would appear that eventually the rocky Pacific coast of
The role of predators in determining the distribution of
organisms in the intertidal and the zonal patterns is best documented for the
Pacific coast of Washington and is discussed here as an example of how complex
biological interactions create the prevailing distributions (Plates 41 and 47).
The dominant abundant intertidal animals on the Pacific coast
other than the space-dominating Mytilus
californianus are the barnacles Balanus
cariosus and Balanus glandula. These
latter two species occur abundantly in the intertidal region even though they
are competitively inferior to M.
californianus, because a predatory starfish, Pisaster ochraceus, preferentially preys on M. californianus, preventing it from completely overgrowing the
barnacles (see Plate 40). Pisaster
ochraceus is a voracious predator of mussels, consuming them at a rate
that prevents them from occupying all the space.
At the same time, Balanus
glandula is found primarily as a band of adults in the high intertidal,
while Balanus cariosus occurs as
scattered, large individuals or clumps in the midintertidal. This pattern, as
Connell (1970) has shown, is also due to predation. Balanus glandula, like Chthamalus
in
It is not known what regulates Nucella populations, but they are preyed on by large Pisaster and are also vulnerable to
periodic events, such as freezing during winter low tides. Such periodic events
may reduce the population by such a significant amount that the Balanus cariosus could successfully
establish themselves in an area. This may explain the differences among various
shores with respect to the abundance of this barnacle.
Although it prefers mussels, Pisaster ocbraceus can consume Nucella
and barnacles of any size and is the primary predator of small- and mediumsized
Mytilus. It is the top predator in
the system. Because of its ability to influence the structure of the entire
intertidal community by consuming Mytilus
and preventing monopolization of the space, it has been called by Paine
(1966) a keystone species as
defined in Chapter 1.
The reason the upper intertidal is a refuge from perdators is
that Pisaster and Nucella can feed only when the tide is
in, and they require a long period to attack their prey successfully. The short
period of immersion of the upper intertidal does not allow sufficient time for
them to make successful forays into that area. In subtidal areas, unlimited
time is available; hence, the starfish have sufficient time to attack and
consume their prey. It is probably for that reason that Mytilus, for example, do not extend into the subtidal areas, even
though they can live there; hence, it is the reason for the sharp lower
boundaries to the zonation of this species (Figure 6.13).
The situation regarding the importance of the interaction of
mussels and starfish in structuring intertidal rocky communities of the Pacific
coast of
intertidal with a
mosaic of patches-some completely dominated by mussels and others with
scattered clumps, depending on the conditions and length of time the dominant
predator was excluded.
Menge (1976) and Lubchenco and Menge (1978) have also
demonstrated that predation is important in setting zonal patterns in the
intertidal of the
An unexplained anomaly of the previous pattern is found in
the very sheltered bays of
The presence of the above mentioned anomalies in the keystone
species concept has caused some researchers to question its usefulness, particularly
how it should be defined and the generality of its occurrence. It is
appropriate to briefly discuss these points.
At its inception by Paine (1969), the keystone species concept
was defined as a single carnivore that preferentially preyed on and controlled
the abundance of a prey species that, in turn, could competitively exclude
other species and so dominate the community. It was subsequently used by
other workers to refer to such
noncarnivorous species as herbivores, mutualists, prey, etc.; hence, confusion
arose. Menge et al. (1994) have redefined the keystone species concept as
originally stated and have clarified it by defining two other predator-prey
relationships that are not keystone relationships. Diffuse predation is a condition where the total predation is
strong and capable of controlling the abundance of a competitively dominant
species but in which the predation is spread over several predators, not just
one. If the total effect of predation on the competitive dominant prey is low such
that predation does not alone control the abundance, the condition is called weak predation.
The large geographical extent of the PisasterMytilus community, extending from
In a recent test of the generality of the keystone concept,
Menge et al. (1994) investigated the high temporal and spatial constancy of the
lower limits of Mytilus distribution
at several sites in
It seems
safe to conclude that the keystone species concept, at least for temperate
rocky shores, remains a valid concept. At the same time, it is not a universal
concept, and its occurrence is variable within the geographic range of the
species or communities in question. It may be replaced under certain
environmental conditions by other community regulatory mechanisms, such as diffuse
or weak predation or physical factors.
A
central theme of marine intertidal ecology, as argued by Connell (1975), is
that wherever predaVon is reduced, competition will be increased. Indeed, the
previous examples demonstrate this. It is well, however, to remember that this
is not the inevitable result of predator removal. For example, Keough and
Data on
the importance of grazing were late in being reported (not until well after
World War II). In the past 20 years, however, considerable evidence has been
accumulated by various methods-experimental removals of grazers, caging
experiments, natural disasters-so our understanding of the role of gazing is
now fairly well advanced. Grazing affects a number of parameters, including
algal zonation, species diversity, patchiness, and succession. Before the
implementation
of various experimental
methods, it was assumed that in the intertidal the upper limits of the dominant
algal species on rocky shores were a function of the species' ability to
tolerate various physical factors. On the other hand, lower limits of algal
species distribution seem not to be set by physical factors, because most algae
grow faster when experimentally transplanted to lower levels or when they are
grown constantly submerged.
Since most algae do grow better when set lower in the
intertidal or submerged, why don't we find them there naturally? The answer coming
from many researchers is that they are grazed out. For example, in various
experiments that have removed grazing animals, the result has been the appearance
lower on the shore of a number of algae otherwise restricted to higher levels,
where grazing is less intense. This has been shown by Lubchenco (1978, 1980)
to be true for Chondrus in
Grazing may also determine the community structure. For
example, Dethier and Duggins (1988) removed the chiton Katharina tunicata from the low intertidal in
scopic scopic plants into a kelp
bed dominated by the kelps Hedopbyllum
sessile, Alaria marginata, and Nereocystis
luetkeana. However, a similar removal of Katharina from a low intertidal area in
Grazing may also set the upper limits of algal distribution.
One of the more interesting pieces of evidence supporting this comes from the
The
mechanisms by which the removal of grazers facilitates the upward or downward
extension of algae vary. In the simplest case, the algae are directly limited
by the grazer. In other cases, the absence of the grazers may permit a rapid
establishment of ephemeral algae, in turn, trapping moisture and permitting
the newly settled sporelings of the dominant algae to survive. In still another
scenario, the upward extension of algae may be seasonal due to changes in
grazing pressure. For example, Castenholz (1961) showed that a summer decrease
in high intertidal diatom cover was due to a summer increase in Littorina grazing activity in contrast
to the less active winter condition. In a more complicated situation, grazing
and physical factors interact. Hay (1979) found that in
Surprisingly enough, some of the limits of algae are set by
the effect bf the algae themselves on potential grazers. In
Another consideration in the zonation of algae species is the
ability of some algae to grow fast enough to escape grazing by becoming too
large for the grazers to feed on. In these cases, the algae will mature only in
areas where the grazers are low in density or restricted from grazing during
some seasons; thus, the algae can reach a size at which they are immune to
grazing.
So far we have considered grazing and its effects on vertical
zonation patterns. Grazing may, however, also affect
the horizontal patterns of zonation-patterns that have as the dominant physical
factor the variation in wave action. The best studied examples come from the
Atlantic coast of
We should not leave this issue of grazing without discussing
a final subject, namely, the defenses of the algae to grazers. We cannot assume
that algae are completely without protection from grazing. Indeed, they are
not. The most obvious adaptations to grazing are morphological, and one of the
most common is the laying down of calcium carbonate in the tissues. Calcium
carbonate reduces the palatability of algae to grazers and makes the grazer
exert more energy in feeding. Furthermore, Steneck (1982) has even shown for
one coralline alga, Clathromorphum, that
the meristem tissue and reproductive sites are all on the underside and
protected from grazing. Growth form may also discourage grazing. Hay (1981) has
shown, for example, that formation of short turfs in algae reduces grazing
mortality. Still other algae, such as the Pacific coast Egregia, have considerable structural or "woody" tissue when
mature; this may deter grazers.
Chemical defenses are also common. Many algae contain noxious
or toxic compounds. For example, on the Pacific coast of North
Finally, there are evolved defenses in the life histories of
certain algae. Some algal species are able to exist in at least two growth
forms, one a low "crust" and the other an upright frondose form. In
areas with heavy grazing pressure, the slow-growing but grazer-resistant crusts
predominate, but in areas of low grazing pressure or in areas from which
grazers have been excluded, the frondose forms increase. Littler and Littler
(1980) have suggested that having these two alternative growth forms in a
single species is a strategy of bet
hedging. The frondose form has fast growth and high competitive ability but
low grazing resistance. The crust form, on the other hand, has slow growth
rates and competitive ability but high grazing resistance. Another life
history feature that may have evolved in algae to cope with grazing is reproduction.
Many algae, particularly those that are favored by grazers because of their
high fraction of digestible material and low fraction of structural tissue,
have evolved the opportunistic habit of high reproductive output. This also
makes them successful in colonizing newly available space. Quick maturity and
high reproductive output mean that these species should be able to colonize an
open area and reach maturity quickly before the grazers find and decimate them.
Other species have evolved the opposite, or K-selected, features. They are
relatively slower growing, perennial, and long-lived. To survive pressure from
grazers, these algae either have evolved chemicals that make them unpalatable
or grow large enough to be immune from grazers. Large size usually also means
an enhanced competitive ability, at least for light.
We can conclude this section on grazing by looking briefly at
the geographical variation in grazing and its effects on various kinds of communities.
Beginning with the temperate zone, grazing is one of the main structuring
agents of the rocky intertidal on the Atlantic shores of Europe and the Pacific
shores of
We can summarize the effects of grazing in the three
intertidal zones defined by the Stephensons to create a general framework for
the North Atlantic Ocean as a model or example of how grazing acts. In the
supralittoral fringe, grazers are either uncommon or have little time to
graze. The rigorous and stressful physical environment is thus the most
important agency causing changes in algal composition. The midlittoral zone is
dominated by barnacles and mussels but is strongly influenced by gastropod
grazers, primarily limpets, and may vary in algal cover depending on exposure.
This, in turn, may limit the numbers of gastropods. Barnacles, however, may
also be present due to the ability of limpets and other grazers to keep the
competing algae in check. In the sublittoral fringe, the algal growth exceeds
the capabilities of the grazers to control it. Therefore, the competition for
space and light among the various algae is the dominant interaction that
structures communities at this level. These various factors and their relative
importance are summarized in Figure 6.17.
The ability of larvae of various benthic invertebrates to
select areas on which to settle has been known for more than half a century
(see Chapter 1). However, the importance of this choice to the eventual
structure of intertidal communities has remained obscure due to the difficulty
of working with larvae under natural conditions. Several studies recently
have, however, demonstrated the importance of this selection ability. In
cal
factors, but by settlement of the larvae.
Furthermore, the barnacle larvae were found by Denley and
Underwood (1979) to avoid settling on substrates already occupied by other
organisms. A more comprehensive example, also from
Larval recruitment can be variable in time and in space. This
variability has been documented, and its importance to the intertidal community
structure can be significant. In some years, any given intertidal area may
experience poor recruitment due to bad combinations of weather, reproduction
by adults, larval mortality in the plankton, such water conditions as wave
action, and so forth. The result is few survivors and a decrease in biological
interactions, such as predation, competition, and grazing. In other years,
favorable physical conditions may swamp an area with recruits, leading to
increased competition or predation and probable changes in the adult population
and the community. As a result, the same area may have a community that not
only differs in composition over time, but may change in relative numbers of
individuals. The importance of competition, predation, and grazing in determining
the structure of the community will also, therefore, change.
Gaines and Roughgarden (1987) have demonstrated another
significant factor that can lead to variation in larval recruitment. They were
able to show that the juvenile rockfish that inhabit the kelp forest
immediately offshore of the intertidal zone in central
This great spatial and temporal fluctuation in the settlement of larvae of the dominant intertidal organisms means that the importance of competition, predation, and grazing are likely to be variable. It is difficult to generalize about the effects of competition, predation, and grazing if the numbers of prey or potential competitors are also unpredictable. Although it is considered by some to
be a "nuisance" in
the setting up of experiments, this irregularity of appearance of various
dominant intertidal invertebrates is a fact of nature and must be considered in
any attempt to understand the structuring process in the intertidal zone. In
other words, the irregular occurrence in a community of a numerically
important, competitively dominant, or keystone predator should lead to a variety
of different outcomes from experimental studies done in the area because of
variations in the intensity of biological interactions resulting from the
larval recruitment patterns.
In an attempt to sort out the relative importance of
competition, predation, and larval recruitment to the structure of rocky
intertidal communities, Menge (1991) undertook a multiple regression analysis
of these factors for New England and
Tide pools
A
characteristic feature of many rocky shores is the presence of tide pools of
various sizes, depths, and locations. Certain conditions affecting life of tide
pools differ markedly from the surrounding intertidal and necessitate a
separate discussion here. Our remarks in this section will be restricted to
those pools that undergo a complete interchange with the ocean water during the
tidal cycle (Figure 6.24). It should be pointed out at this juncture that the
biotic communities of tide pools and the factors influencing their structure
are less well understood and studied than those of the emergent rocky
surfaces. It has even been suggested that tide pools do not represent an
intertidal habitat because they are never exposed to the air during a tidal
cycle. However, the fluctuations in physical and chemical factors in tide pools
are a function of the tidal cycle; hence, it seems logical to consider them in
this chapter. Furthermore, many of the organisms found in tidal pools are
similar to those found on the adjacent exposed rock.
At first glance, tide pools appear more or less ideal places
for aquatic organisms seeking to escape the harshness of the intertidal during
its exposure to air. In reality, however, escape from such physical factors as
desiccation may mean exposure to others that operate more severely in tide
pools.
Tide pools vary a great deal in size and in the volume of
water they contain. Since water is a great moderator of harsh physical
conditions, the larger the pool and the greater the water volume, the less the
fluctuations in physical factors. Other factors in addition to volume influence
the physical and chemical conditions of the water held in a tide pool. These
include the surface area, depth of the pool, height in the intertidal, exposure
to wave action and subsequent splash, degree of shading, and drainage pattern.
In addition, the physical and chemical environment of tide pools can vary vertically
with depth and horizontally across the pool. Finally, all physical factors may
fluctuate diurnally and seasonally. Given all these variables, Metaxas and
Scheibling (1993) suggest that it is unlikely that any two tide pools will be
similar in all characteristics; therefore, individual pools are unique physically.
If this is true, it means ecologists cannot replicate experimental
manipulations in different tide pools and may be a significant reason why there
has been so little experimental ecological work done on pools. It may also be
the reason why most studies have been primarily descriptive.
Three major physical factors are subject to
variation in tide pools. The first is temperature. Whereas the ocean itself is
a vast reservoir that heats and cools very slowly and usually within very
narrow limits, the same is not true of tide pools. These relatively small
bodies of water are subject to more rapid changes. Shallow tide pools exposed
to the sun on warm days may quickly reach lethal or near lethal temperatures. Similarly,
tide pools in cold temperate or subpolar regions may have temperatures in the
freezing range in winter. An additional problem is temperature fldctuation.
The pool may either heat up or cool down over a several-hour period while
exposed to the air, but when the tide returns, it will be flooded with ocean
water. This will suddenly change the temperature of the whole pool. Variations
in daily temperature in a tide pool can be as much as 15°C,
depending on its height in the intertidal, volume, degree of shading, and wave
exposure. Finally, change in temperature during exposure at low tide may cause
temporary thermal stratification of the pool. Organisms inhabiting such pools
must still be adapted to considerable temperature fluctuation.
The second factor to vary in pools is
salinity. During exposure at low tide, tide pools may heat up, and evaporation
occurs, increasing salinity. Under hot tropical conditions, the salinity
increase can be dramatic enough to reach the point of precipitating out salt.
The opposite situation is the case when heavy rains occur at low tide and flood
pools with fresh water, dramatically lowering the salinity.
Fluctuations in salinity in tidal pools vary with the position of the pool on
the shore and the other factors mentioned, but values that have been measured
have ranged between 5-25 psu. Salinity stratification also may develop in the
pool due to fresh water runoff during heavy rains or as a result of freezing in
winter and evaporation in summer. Again, tide pool animals and plants may have
to be adapted to wider ranges in salinity than typical marine or intertidal
organisms. As before, when the tide returns, the pool will be flooded with
seawater at some point, and there will be an abrupt return to normal
conditions.
The final physical factor undergoing change in
pools is oxygen concentration. Since the amount of oxygen that can be held in
seawater is a function of temperature, it follows that tide pools that heat up
during exposure to the air will lose oxygen. Under normal conditions, this may
not be serious enough to produce oxygen stress, but if the pool is crowded with
organisms, it may produce a stress situation. For example, a pool filled with
algae that was exposed at night would produce a situation in which the lack of
photosynthesis coupled with high respiration could reduce the oxygen level significantly.
Oxygen level has been recorded falling to only 18% of saturation in tropical
tide pools. It is also possible to develop an oxygen
stratification in the water column of certain pools.
Tide pools are areas of refuge from
desiccation for intertidal organisms, but in turn, these organisms suffer from
rapid changes in temperature, salinity, and occasionally oxygen; thus, the
fauna and flora is restricted to those organisms able to tolerate such ranges.
The
organisms that inhabit tide pools are similar to those on the adjacent emergent
substrates but often with differences in abundances between the pools and
exposed surfaces. For example, several genera of algae, such as Spongomorpha and Corallina in the intertidal of
Zonation patterns have been described for tide pool organisms
both within the intertidal zone and vertically within the tide pools. Along the
intertidal gradient such green algae as the genera
Enteromorpha, Cladophora, and Chaetomorpha,
dominate the upper tide pools,
while such brown algae as Fucus and Laminaria, and the red corallines, such
as Lithothamnion and Corallina, are abundant in the lower
intertidal pools. Benthic invertebrates also show a zonation. High pools are
inhabited by littorine snails, whereas low tide pools have a greater diversity
of snails and all invertebrates. Fish zonation in tide pools has not been
quantitatively documented according to Metaxas and Scheibling (1993), but
qualitatively, there is a decrease in the number of species with increasing
height of the pool. In general, both in number of species and biomass, high
tide pools are depauperate compared with lower level pools.
The role of biological interactions in determining the
community structure of tide pools is poorly documented when compared to the
studies for the exposed rocky intertidal. This is due not only to a relative
lack of studies, but also to the variability among adjacent tide pools at the same
intertidal level when subjected to experimental manipulation. Such variability
has precluded obtaining statistically significant results in many cases,
although correlative data often indicate a trend. We discuss some of these data
here.
Herbivory has similar effects in tide pools to those recorded
for the emergent rock surfaces, namely, the altering of macroalgae abundance.
For example, Paine and Vadas (1969) demonstrated that the removal of sea urchin
grazers from tide pools in
Very few studies have investigated the role of predation in
structuring tide pool communities. Indeed, there seem to be no well-documented
manipulative studies. The few studies that have been done suggest that
addition of predators to pools reduces the abundances of various prey
organisms, but whether or not the predator exerts control of the community
structure is uncertain.
The evidence for the importance of interspecific competition
in regulating community structure in tide pools is also sparse. Lubchenco
(1982) and Chapman (1990) have both demonstrated decreases in canopy cover in Fucus due to competition with ephemeral
algae. Kooistra et
al. (1989) have shown competitive dominance as
indicated by overgrowth for the alga Halichondria
panicea in tide pools in Brittany,
Although recruitment can be a significant factor in
structuring tide pool communities, no studies have considered this factor
directly. Similarly, there is little or no information about the role of
disturbances, particularly large-scale episodic events, in the structuring of
tide pool communities.
Marine
Science Name___________Pd___
Rocky Shores Reading Part 1
- How do
rocky shores compare with sand and mud shores?
- What types
of animals dominant the intertidal rocks?
- What types
of animals dominant the subtidal rocks?
- What is the
most striking feature of any rocky shore?
- How is one
zone different from another?
- Compare and
contrast rocky shores with mountains?
- What are 3
ways rocky intertidal zones vary?
a)
b)
c)
- What
conditions may alter the bands or zones?
- In the
Stephenson’s scheme, there are three main divisions. What are they?
10
Describe each zone discussed in question 9
Physical
Factors-that cause zonation
11 What does desiccation mean?