WORM CRUSTACEAN ECHINODERM READINGS
1. Sea moss may yield 'designer' therapies to attack human cancers
|
|
March 28, 2000Web posted at: 11:58 a.m. EST (1658 GMT)From staff reports
(CNN) -- Sea moss growing on rocks at the bottom of oceans may contain chemicals allowing scientists to make "designer" medicines that precisely target cancers.
Studying compounds from bryozoans and other life on or near coral reefs has occupied medical scientists for more than three decades, and now Dr. John Marshall of Georgetown University Medical Center reports particular promise with a marine-organism extract, "briostatin."
When used with traditional chemotherapies, briosatin can help customize treatments by modulating chemical signals within a cancerous cell to make the cell more nearly normal, he said.
"We are moving to a time where we are no longer going to treat cancer patients based on just what kind of cancer they have," Marshall said. "We won't treat all colon cancer patients the same. We won't treat all lung patients the same.
"Where we are moving to is taking an individual's cancer and measuring particular characteristics of it and saying, 'OK, you've got this wrong, this wrong and this wrong in your tumor and, therefore, I am going to treat you with Drug X, Drug Y and Drug Z because that is tailored to your cancer.'"
Marshall has used briosatin by itself and with other chemotherapy drugs in about 60 patients. The results have been "significant anti-cancer activity," plus briostatin does not cause hair loss.
Alone, the extract is not "powerful enough to be an answer for many cancer patients," he said. "I'd like to be proven wrong on that, (but) our hopes are that it will bring to chemotherapy enough additional power to help them significantly."
While briostatin does not prompt hair loss, it does produce increased muscle ache and fatique, Marshall said.
More than 30 other drugs derived from the ocean are being investigated by medical researchers. The ingredients for one drug, "squalomin," comes from the bottom-dwelling dog shark (also called dogfish and grayfish). This chemical has the ability to destroy blood cells necessary to feed tumors, thus starving the cancer cells.
CNN Medical Correspondent Eileen O'Connor contributed to this report.
2. TUBE WORMS
Scientists have discovered bacteria living on the sea floor that don't need light to live. It's thought that these bacteria originate below the sea floor, where they live under very high temperatures and pressures. It's also thought that they make up more living matter than do all of the other living creatures on Earth combined -- in the water and on land.
Dr. Stephen Hammond of the VENTS program of the National Oceanic and Atmospheric Administration described the discovery of these bacteria in the 1970s as comparable to finding an undiscovered tropical rain forest. He said there's just as much diversity of species among the bacteria.
The bacteria form the foundation of an entire ocean ecosystem. They live inside the tissues of giant tube worms -- which can grow to nearly three meters long. Fish eat the worms, and, in turn, become food for larger fishes and so on. The worms are filled with the bacteria. There are more bacteria living in just one ounce of worm tissue than there are people living on Earth. The worms and the bacteria help feed each other. A worm's bloodstream carries nutrients to the bacteria. In return, the bacteria create waste products that feed the worm.1
Introduction to the Pogonophora Weird tube worms of the deepest seas
In 1900, a strange tube-dwelling worm was dredged from deep waters around Indonesia. While somewhat resembling tube-dwelling annelids, it lacked obvious segmentation; even more strangely, it also lacked a mouth, gut, or anus. This was the first discovery of the Pogonophora, an animal phylum restricted to the deep sea and remarkably common in certain habitats there.
About 80 pogonophoran species are known today, with new species still being discovered. One of the most spectacular zoological discoveries of recent years was the finding in 1977 of giant pogonophoran worms, 1.5 meters long, growing in heated, sulfur-rich water around warm-water vents in the Pacific Ocean, 2600 meters below the surface (pictured at right). These worms are sometimes placed in their own phylum, the Vestimentifera, but they are similar to pogonophorans in most respects, and the current tendency is to group these rift-dwelling worms together with the rest of the Pogonophora into one phylum.
The name Pogonophora is Greek for "beard-bearers," and comes from the fact that many species have from one to many tentacles at the anterior end. These tentacles somewhat resemble the lophophore found in animals like brachiopods and bryozoans, as well as the feeding tentacles of certain chordates. The incompletely known anatomy of pogonophorans was interpreted to show that pogonophorans were chordate relatives. Because pogonophorans live with their lower ends buried in mud, and were broken during the dredging process, it was not until 1964 that a complete pogonophoran was recovered. It turned out that pogonophorans have a segmented posterior end of the body -- the opisthosoma -- that bears setae and resembles an annelid body. The forward part of the body, or prosoma, is unsegmented. Because of the segmented opisthosoma, and because pogonophoran larvae have been found to look very much like annelid larvae, pogonophorans are now considered to be close relatives of the annelids.
How do pogonophorans feed with no mouth or gut? Some nutrition is provided by absorbing nutrients directly from the water with the tentacles. But most of a pogonophoran's nutrition is provided by symbiotic bacteria living inside the worm, in a specialized organ known as the trophosome that develops from the embryonic gut. Inside the trophosome, these bacteria oxidize sulfur-containing compounds such as hydrogen sulfide, which pogonophorans absorb through their tentacles -- the bright red color of rift-dwelling pogonophoran tentacles is due to hemoglobin, which absorbs both sulfides and oxygen for the use of the bacteria. The bacteria derive energy from sulfur oxidation, which they use to fix carbon into larger organic molecules, on which the pogonophoran feeds.
The fossil record of pogonophorans may extend back to the Vendian Period; long thin tubes known as sabelliditids have been found in rocks of that age, and somewhat resemble pogonophoran tubes. However, studies on sabelliditid structure have proved inconclusive in determining exactly what these fossils were. A few fossil pogonophoran-like tubes have turned up in later deposits (e.g. Adegoke 1967), but pogonophorans are generally quite rare as fossils. 2
Creatures of the Thermal Vents
by Dawn Stover
The three-person submersible Alvin sank through the cold, dark waters of the Pacific Ocean for more than an hour, finally touching down on the sea floor more than 8,000 feet below the surface. It was December 1993, and the scientists inside the sub had come to this stretch of the East Pacific Rise, an underwater mountain range about 500 miles southwest of Acapulco, Mexico, to inspect a recently formed hydrothermal vent - a fissure in the ocean bottom that leaks scalding, acidic water.
Peering out through the sub's tiny windows, the visitors were astonished to seethickets of giant tube worms, some four feet tall. The tail ends of the worms werefirmly planted on the ocean floor, while red plumes on the other ends swayed like afield of poppies. Alvin had brought researchers to the same spot less than twoyears earlier, when they had seen none of these strange creatures. Measurements atthe site have since shown that individual tube worms can increase in length at a rateof more than 33 inches per year, making them the fastest-growing marine invertebrates. That means tube worms can colonize a vent more rapidly than scientists once thought.
The giant tube worm is one of the most conspicuous members of a diverse community that forms around hydrothermal vents. Scientists once thought that no living thing could survive the harsh combination of toxic chemicals, high temperatures, high pressures, and total darkness at these vents. But in 1977, researchers diving in Alvin discovered tube worms and other bizarre organisms thriving at a vent off the Galapagos Islands. Similar communities have since been found at several hundred hot spots around the world. These creatures are like nothing else on Earth.
Vents form where the planet's crustal plates are slowly spreading apart and magma is welling up from below to form mountain ranges known as mid-ocean ridges. As cracks form at these spreading centers, seawater seeps a mile or two down into the hot rock. Enriched with minerals leached from the rock, the water heats and rises to the ocean floor to form a vent.
Vents are usually clustered in fields, underwater versions of Yellowstone's geyser basins. Individual vent openings typically range from less than a half inch to more than six feet in diameter. Such fields are normally found at a depth of more than a mile. Most have been discovered along the crest of the Mid-Oceanic Ridge, a 46,000- mile-long chain of mountains that wraps around Earth like the seams on a baseball. A few vents have also been found at seamounts, underwater volcanoes that are not located at the intersection of crustal plates.
The largest vent field, called TAG (short for Trans-Atlantic Geotraverse), is about the size and shape of a football stadium. Other fields have more whimsical names like Clam Acres, Mussel Bed, Rose Garden, Garden of Eden, Broken Spur, and Lucky Strike. Snow Blower is named for the white, flaky bacteria discharged from its vents. Genesis is a vent that sputtered out but came back to life a few years later.
Hydrothermal vents are underwater oases, providing habitat for many creatures that are not found anywhere else in the ocean. More than 300 new species have been identified since the first vent was discovered in 1977.
Besides the giant tube worms, which have so far been found only in the Pacific, there are pencil-size Jericho worms with accordion-like tubes; orange worms covered with tiny bristles; small benthic worms that wriggle through the mud; and finger-length, dark red palm worms that stand upright, topped with wiglike fronds. A special class of small worms, called Alvinellids (named after the sub), live on the walls of mineral deposits that form around vents.
Mussels, shrimp, clams, and crabs are abundant at many vents, but these are not the same species that you find in seafood dishes. The cocktail-size shrimp that dominate vents in the mid-Atlantic, for example, have no eyes. However, at least one species has an extremely sensitive receptor on its head that may be used to detect heat or even dim light coming from vents. Scientists still aren't sure how shrimp and other vent creatures cope with chemical-laden seawater that would kill ordinary shellfish.
Biologists have observed a variety of smaller crustaceans around vents, including miniature lobsters called galatheids, and amphipods resembling sand fleas. They have also seen snail-like limpets the size of BBs, sea anemones, snakelike fish with bulging eyes, and even octopuses.
While octopuses are at the upper end of the vent's food chain, bacteria are at the bottom. They are the first organisms to colonize newly formed vents, arriving in a snowlike flurry and then settling to form white mats or tendrils attached to the ocean floor. Bacteria have been found living beneath the ocean's floor, and it seems likely that they emerge from below when the conditions are right. Vent bacteria can withstand higher temperatures than any other organism. That makes them attractive to researchers who are developing heat-stable enzymes for genetic engineering, and culturing bacteria designed to break down toxic waste.
Water pouring out of vents can reach temperatures up to about 400 C; the high pressure keeps the water from boiling. However, the intense heat is limited to a small area. Within less than an inch of the vent opening, the water temperature drops to 2 C, the ambient temperature of deep seawater. Most of the creatures that congregate around vents live at temperatures just above freezing. Thus chemicals are the key to vent life, not heat.
The most prevalent chemical dissolved in vent water is hydrogen sulfide, which smells like rotten eggs. This chemical is produced when seawater reacts with sulfate in the rocks below the ocean floor. Vent bacteria use hydrogen sulfide as their energy source instead of sunlight. The bacteria in turn sustain larger organisms in the vent community.
The clams, mussels, tube worms, and other creatures at the vent have a symbiotic relationship with bacteria. The giant tube worms, for example, have no digestive system - no mouth or gut. "The worm depends virtually solely on the bacteria for its nutrition," says microbial ecologist Colleen M. Cavanaugh of Harvard University. "Both partners benefit."
The brown, spongy tissue filling the inside of a tube worm is packed with bacteria - about 285 billion bacteria per ounce of tissue. "It's essentially a bacterial culture," says Cavanaugh.
The plumes at the top of the worm's body are red because they are filled with blood, which contains hemoglobin that binds hydrogen sulfide and transports it to the bacteria housed inside the worm. In return, the bacteria oxidize the hydrogen sulfide and convert carbon dioxide into carbon compounds that nourish the worm.
Tube worms reproduce by spawning: They release sperm and eggs, which combine in the water to create a new worm. Biologists don't know how the infant worm acquires its own bacteria. Perhaps the egg comes with a starter set.
Scientists also don't know how tube worms and other organisms locate new vents for colonization. "The vents are small, and they're separated, like islands," says Cindy Lee Van Dover, a biologist and Alvin pilot who studies vent life. Most vent organisms have a free- swimming larval stage. But scientists aren't sure whether the larvae float aimlessly or purposely follow clues - such as chemical traces in the water - to find new homes.
Studying the life cycle of vent organisms is difficult. Researchers have visited only a fraction of the ocean's hot spots. They have been able to observe vent life only by shining bright lights on creatures accustomed to inky darkness, and many specimens die quickly when removed from their unique environment. Underwater cameras are helping scientists make less intrusive observations, but diving expeditions are still the most useful way to gather information. The 1993 Alvin expedition to the East Pacific Rise was one in a series of dives to the area. The site was first visited in 1989, and scientists observed vent organisms thriving there. But when Alvin returned in April 1991, its flabbergasted occupants witnessed the birth of a hydrothermal vent. A recent volcanic eruption had spread glassy lava across the ocean floor, and the researchers measured temperatures up to 403 C - the hottest ever recorded at a hydrothermal vent. The scientists dubbed the site Tube Worm Barbecue, because the worms they brought back to their ship had charred flesh.
"The most spectacular sight down there was this massive blinding snowstorm of bacteria," says Rich Lutz, a marine ecologist at Rutgers University, who led the expedition. On the ocean floor, the bacteria formed mats several inches thick, but the scientists saw no other living things.
Since the eruption, scientists have been able to watch several stages of colonization at the site. When they returned in March 1992, only a few bacterial mats remained. In their place were colonies of Jericho worms and a variety of small crustaceans. The scientists named the area Phoenix, because new life had arisen from the ashes of the eruption.
The scientists first observed the giant tube worms at Phoenix in December 1993. They also noticed a number of mineral deposits, some towering to heights of more than 30 feet. These structures form where hot vent water meets cold seawater, causing metal sulfides to precipitate out. The precipitating sulfides, which look like smoke, amass to form chimneys called black smokers. Like the vent fields, some smokers have names. Smoke and Mirrors, for example, has shelflike overhangs that trap hot water rising from below, creating upside-down shimmering pools. The largest known black smoker is Godzilla, a 160-foot-tall structure off the coast of Oregon.
During a December 1993 dive to the Phoenix vent field, Alvin accidentally toppled a 33-foot-tall smoker. When the sub returned for a brief visit three months later, the chimney had already grown back 20 feet. Scientists were surprised by the speedy recovery, which seems to parallel the rapid growth of tube worms and other organisms at the vents. The visits to the Phoenix site "give us a sense of how quickly these vents are colonized," says Van Dover.
Another expedition is planned for November. By then, the community of organisms now prospering at the vents may already be a ghost town. When the flow of hot, sulfide-rich water slows to a trickle, death also comes quickly. 3
3. BRACHIOPODS:
Ancient Dwellers of the Sea
by DANIEL R. TARDONA
Tardona is a naturalist with the National Park Service at Great Smoky Mountains National
Park He has researched fossil brachiopodsand marine mammals. He took all the photos.
One of the most abundant and diverse forms of life ever to live in the oceans of the earth is a group of shelled animals called brachiopods. Yet it seems that seashore naturalists are only remotely acquainted with these fascinating creatures. While it is unlikely that the average beach walker will find the shell remains of this interesting marine animal, paleontologists commonly find fossilized shells of these ancient sea inhabitants. Despite there being only about 300 species of these animals in existence today, it is estimated that over 30,000 different species have inhabited the world’s oceans over the last 570 million years.
Brachiopods are certainly a very adaptive, hence successful, group of animals. Just what is a brachiopod, or lamp shell, as it is sometimes called? (The shape of the two shells of some species [for example, Magellania spp.1 resembles a spout at the apex, reminiscent of a Roman lamp.) This two-valved marine invertebrate looks very much like a clam, but is not part of the group of animals that includes clams, oysters, squids and the like (phylum Mollusca). Regardless of their superficial molluscan looks,
brachiopods are classified into their very own phylum: Brachiopoda. They are solitary, bivalves (two shells), and bilaterally symmetrical. The shells are of unequal size, a characteristic key to distinguishing brachiopod shells from clams and mussels, which have shells of equal size. But to really understand the differences between the mollusks and the brachiopods we need to look deeper.
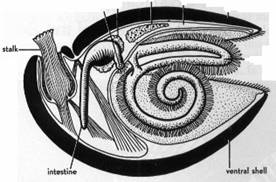
Brachiopods have a very different ar rangement of internal organs. The most striking part of the internal anatomy is a ciliated, filament bearing structure called the lophophore. In some ways it resembles a convoluted, hairy pipe cleaner. This structure occupies much of the internal cavity of all living brachiopods and is the unifying feature of the phylum. The lophophore functions as the principle food gathering and respiratory organ of this animal, as well as an organ for excretion. The constituent filaments of the lophophore are controlled individually. Thus, in food gathering it works as a filter. It is an open filter not readily clogged like the gill of the clam. The brachiopod can allow objectionable, unwanted particles to pass through. As you may have guessed, brachiopods are suspension feeders which means they ingest minute particles of food suspended in the water. The lophophore pumps water through the shell and extracts nutrients and oxygen from it. The lophophore is in part the reason this living fossil has been so successful, allowing the brachiopod to tolerate turbidity, more so than the clam with its siphon system.
Brachiopods can be broken into two large groups or classes: the articulates and the inarticulates. Articulates, as the name implies, are brachiopod species whose shells (or valves) are hinged together by a set of interlocking teeth and sockets. The inarticulates are those species that lack the hinge arrangement. The valves of the inarticulates are held together by muscles. There are four orders of inarticulate brachiopods, most of which have been around a very long time. For example, the order Lingulida has existed for over half a billion years. Lingula in this order is a burrowing inarticulate brachiopod which has remained in a relatively unchanged state since the Cambrian period, and today lives in a brackish to intertidal environment. Inarticulate brachiopods are usually considered less advanced, less diverse, and less abundant than the articulate brachiopods.
There are numerous differences between these two classes of brachiopods, most occurring during the larval stage and involving locomotion, feeding, and development of a stalk-like appendage called the pedicle. The pedicle is another feature of brachiopods that contributes to the uniqueness of these animals. Li,zgula, for example, lives in a vertical position in a species today have a pedicle, but this appendage is lost in other species. There is a great diversity of pedicle forms, and they perform a variety of functions. The pedicle helps some species to attach to the sea floor or some other substrate. In some cases the pedicle can be used to lift and rotate the shell. Some species of brachiopods attach with cementation similar to barnacles; some attach by spines; and some species today, such as the genus Thecidellina, even have byssus-like threads like mussels.
Adaptations of the pedicle, lophophore, and various other physiological strategies have helped brachiopods live on different substrates, and to survive in environments with varying temperatures, sea depths, and water current strengths. A diversity of shell shapes and structures has likely contributed to the longevity of the brachiopod. Different shell designs may function to stabilize the brachiopod, and the ability to stabilize may allow some species to live in turbulent environments. Perhaps a broad, flat shape that can be observed in some fossil forms (such as Straphoinenids) may have helped some brachiopods avoid sinking
in fine sediments. Still other factors, like the thickness of the shells, the amount and coarseness of the ribbing on shells, or other “ornamentations,”may dictate the strength of the shells, making them resistent to attacks by shell-crushing predators. As you might guess, few shell designs, if any, would be absolutely predator-proof. Some articulate brachiopods, however, have evolved an interesting strategy for warding off predators. The flesh of some species has a noxious smell and taste, a biochemical defense against shell drillers and crushers.
Obviously, there is much to be said about these fascinating, ancient dwellers of the sea. This brief introduction only touches on the great diversity displayed through geologic time by these adaptable sea creatures. While the heyday of the brachiopod may now be past, today they are more common and inhabit more kinds of environments than was thought only a few years ago. Living species can be found in the seas of Jamaica, Australia, New Zealand, the San Juan Islands, the Antarctic, and the Arctic, to mention a few. There is still much to be learned about fossil species, and even more about today’s living brachiopods. Certainly, this ancient sea dweller may continue to thrive for a very long time.
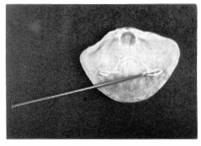 The
brachial valve of the extant articulate brachiopod,
Terebratulalia transversa.
The
brachial valve of the extant articulate brachiopod,
Terebratulalia transversa.



An extant articulate brachiopod showing the opening through which the pedicle passes.
4. Along Came a Spider
By Joseph Albanese
UN Vol. 20 No. 2 1991
I enjoy SCUBA diving the rocky shorelines, jettys and inlets of the middle Atlantic coast especially in the fall when water temperatures warm into the mid-sixties and bring tropical fish out of Gulf Stream eddies. Here in the littoral zone they’ll find some shelter from predators but not from approaching winter’s cold.
It was during August of 1989, while diving at a place called Land’s End on the eastern tip of Newport, Rhode Island, that I came upon an unusual creature. At least it was unfamiliar to me. It’s motion along the bottom was almost unperceptible. I gently picked it up in my gloved hand for a closer examination. I decided it looked a lot like a spider and let it go.
I wasn’t certain that there were spiders living in the ocean 25 feet below the surface. So, when! got home, I opened Kenneth Gosner’s excellent Field Guide to the Atlantic Seashore. Sure enough there are drawings and descriptions of seven species of sea spiders that might be found in these waters. These spiders are anatomically different from terrestrial ones. For one thing they do not have an obvious abdomen or opisthosoma body segment. Their internal organs are arranged in their legs. There are other more subtle anatomical differences between the two.
When! returned to Land’s End in September 1 was ready with my Nikonos camera and 1:1 extension tube to find another sea spider and take its picture. This dive I found not one but two spiders.
So, how come I never noticed these things before? Well, for one thing, they aren’t very big. These inshore species are only about 114 inch. Deep water species are bigger; some with leg spans of 20
inches. Sea spiders also blend-in very well with their surroundings.
According to Gosner they take on the color of their surroundings. I doubt this means they have the ability to change coloration the way a flounder or chameleon can. The reason probably has more to do with diet. As Rachel Carson says in The Edge of the Sea, “For some reason that may have to do with its food, it is the same apricot color as the bryozoan carpet beneath it; the sea spider, too, seems the embodiment of fragility.” Either way my specimen was more black than any color.
Luckily, one photograph was at least good enough to identify the species I’d seen. I compared my developed 35mm slide to the drawings and descriptions in Gosner’s book, and as near as I could tell I’d photographed a lentil sea spider, Anoplodactylus lentus. The normal range for the lentil sea spider is from Cape Cod south to the Caribbean. And I’ve seen on television that other species even live under the Antarctic ice cap. Sea spiders are often found with hydroids (that’s where I found mine) and presumably feed on them.
Alice and Robert Lippson confirm that sea spiders are found among hydroids and other epifaunal growths in their book Life in the Chesapeake Bay. In the bay they are found in water of 11 to 30 parts per thousand salinity.
Next time you dive off the Atlantic Coast look closely at the growth along the bottom. You might be alongside a spider. Next time I see one I may just breath up an entire cylinder of compressed air watching their behavior to see if they eat some hydroids or change color. Well, maybe not. There are lots of other things to see down there, you know.

5. “Respect for an Old-timer” Horseshoe Crab
by John R. Quinn
It looks something like a cross between Sherman tank and a vacuum cleaner. It’s as heavily armored as a medieval knight and just about as clumsy in movement. It plods its way resolutely through life, oblivious, for the most part, to everything around it and secure in its invulnerability to all that a hostile world can throw at it.
It looks dangerous, indeed venomous, but it is in fact a totally harmless animal. It is superbly equipped to cope with all of the threats and stresses of its surroundings save for one. It is utterly defenseless against the sticks and stones of man. It is an ancient, living paradox whose roots predate the dinosaurs. It’s eventual ending may, if fate so decrees it, supersede humanity itself.
It is Limulus polyphemus, more popularly known as the horseshoe crab. It’s hard to imagine a more misunderstood and unjustly feared and reviled creature of the shore environment than Limulus.
One of the most distressing sights along the back-bay beaches of the New Jersey shore in summer is the battered and broken carcasses of numerous horseshoes. They’re found bludgeoned to death by rock- or stick-wielding beach walkers. These humans take advantage of the gentle creatures’ considerable vulnerability during the breeding season. These beach walkers often indulge in that all-too-familiar human penchant for destroying something they cannot or will not come to understand. These lumbering creatures were assigned their common name by folks who assumed that because they have a shell like a crab, legs like a crab, and live in the marine environment like a crab, they must be a crab. But in fact Limulus is much more closely related to the earthly spiders and scorpions than to any crab.
It is an arthropod (eight-legged), an arachnid, to be scientific about it. They look as though they might better belong in the abyssal depths of the sea. But horseshoe crabs are basically shallow water animals inhabiting the subtidal zone down to about 75 feet of depth. Compared to these creatures, man is a mere upstart, a callow newcomer to the game of life.
Ancestral horseshoe crabs first saw the light of the primeval day back in the Devonian period of the earth’s history some 350 million years ago. The species Limulus has remained virtually unchanged since the earliest days of the dinosaurs, or for about 200 million years. Such a long lineage has obviously served Limulus well in coping with life. After all, they’re still around, even in the face of growing marine pollution and human persecution throughout much of their range. This extends from about Bar Harbor, Maine, to the Gulf of Mexico, with related species further south. But such geological longevity, awesome though it may seem, could not possible have prepared Limulus for cohabitation with people in the twilight of existence. The creature is simply too big and striking to escape notice, particularly when it must come ashore in considerable numbers to reproduce its kind. It is at this time, normally from mid-May to early July, that the majority of human-horseshoe encounters occur. The outcome of most of them is something less than pleasant for the horseshoe crab.
Most of the horseshoe crab’s mystique (and the resultant fear reaction among humans) stems fro two major aspects of the animal’s physique and conduct. Its tail spike, or telson, looks like a “stinger” to the initiated. The horseshoe’s slow, deliberate movements and total lack of a flight response suggest a confident willingness to use it if handled. The closest most uninformed beach walkers will come to interaction with a horseshoe crab is to nudge it with a foot, poke it with a handy stick, or worse. Few people, even grown men, will willingly handle one. Most are convinced the creature is dangerous and will inflict grave injury if given the chance.
Shore residents and visitors most often meet the horseshoe crab during its spring and early summer breeding season. This is when the animals approach the beaches of quieter bays and estuaries to mate and deposit their numerous eggs in the sand.
At this time they can be observed on the high tides in the shallows of such bodies as Barnegat and Great Bays and the bay side of Long Beach Island. They are quiet common in the slat marsh areas of Island Beach State Park.
The female Limulus is an impressive animal, reaching a length of about two feet(including tail) at maturity. The males are always smaller. Small groups of them actively pursue their would-be mates over the sandy shallows. Eventually one will gain a claw hold on the female’s back for the final trip to the beach and the actual egg laying.
During spawning the pair will burrow into the sand and a large female may deposit up to a thousand eggs, burying them several inches in the wet sand at the high tide mark. There they are immediately fertilized by the male. The wave wash covers them with more sand. If all goes well, the eggs hatch in about three weeks’ time. The tiny young make perfect miniatures of the adults minus the tail. This look changes with the first molt when they make their perilous way to the water on the next high tide and to their destiny in life.
In spite of all its energetic wriggling and flexing when handled, which give the impression of strength and menace, the largest horseshoe crab is as harmless as a beach flea. A cursory look at its anatomy will clearly depict that fact. To begin with, the telson is simply a hard, horny appendage employed as a balancing organ and as a lever when the horseshoe gets turned over on its back. It is incapable of delivering a real sting. For most larger individuals contact is at best no sharper than a blunt pencil point.
The animal’s five pairs of walking legs are weakly clawed and designed solely for moving its ungainly body across the sand and mud bottoms it prefers. It probes the mud for the small marine life and vegetation it eats.
The primitive mouth opens deep within the base of the legs. It has no “lips” or appendages as such. It serves simply as a grinding mill for whatever food the small claws pass on to it. The only other appendages the horseshoe possesses are its unique “book lungs” or gills–its breathing apparatus. These, in five pairs, are located just behind the legs near the tail and overlap each other like the pages of a book–hence the name. These are the most delicate and vulnerable part of the creature’s anatomy, and a horseshoe stranded on its back on a beach is in a very precarious position indeed. Gulls will quickly notice its plight and gather to feed on the soft, leaflike gills. This is about the only part of Limulus that’s edible to all but the most determined, hungriest predators.
Years ago, when horseshoe crabs were more abundant than they are now, they were collected in sizable numbers and chopped up for fertilizer. Today, about the only practical use to which man puts the armored creatures is lobster trap bait. This occurs, thought, only on a limited, local scale.
A rather clever device looking something like a primitive paper cutter was invented for bait preparation. The hapless horseshoe was placed upside down on the gadget and cleaved in half (alive, or course) by a large, levered blade.
The shed, sun-dried carapaces of the growing young are often picked up by beachcombers and shellacked as home decorations. Small versions sometimes turn up in the marine section of local pet shops. Although they are quite hardy, horseshoes will not thrive in the marine aquarium unless they are offered a variety of animal and vegetable foods in the form of brine shrimp and algae. They should be kept with smaller, more placid tank mates. Most larger fish will harass and pick at them. The horseshoe crab shares with the extinct dodo the unfortunate renown of being a creature suited to its original environment but being totally unable to adapt to the most recent appearance of people is it. In this respect, it is the polar opposite of animals like rats, roaches, and herring gulls. These creatures have not only learned how to coexist with and yet avoid man. They have actually profited from our construction, agriculture and, of course, our waste products.
Horseshoe crabs gain nothing from civiliza-tion and in fact cannot deal with it or adapt to its heavy impact in any effective way. In the end, these highly specialized and woefully inflexible creatures depend utterly on the mercies of the conqueror for their continued survival, if it is to be granted at all.
There’s not much of value in a horseshoe crab, so their problems and decline today cannot be attributed to over-harvesting for food or deliberate hunting for sport. Rather, it’s a casual destruction. Limulus must endure–the failing health of the marine environment in general
6. These blue bloods save lives-- now theirs are in danger
Published in The Orlando Sentinel on September 10, 2000
steve helber/associated press Rack of crabs. Horseshoe crabs bleed into bottles at a lab in Chincoteague Island, Va. The blue blood is the only source of a compound used to test for impurities in drugs, vaccines, artificial limbs and IV drips. `These are the most incredibly resilient, tough, fascinating creatures.` Jim Berkson Virginia Tech assistant professor
Horseshoe crabs make a substance to test for contaminants, but the species may be waning.associated press
CHINCOTEAGUE ISLAND, Va. -- In a small unmarked building on a back island road four miles off the coast of Virginia, the bleeding begins.
Hundreds of strange, spiked creatures are pinned to plastic racks as lab technicians in white coats and face masks glide from row to row, rubbing them with alcohol as though soothing them for their ordeal.
The creatures flinch as long surgical needles are inserted into creases in their helmet-shaped shells. Blood spurts into bottles beneath them.
Rich and blue, it is precious as gold.
And every day it saves lives around the world.
No other creature can produce this substance, the world`s only known source of a compound used to test for contaminants in every drug and vaccine, every artificial limb and every intravenous drip in every hospital in America. Science can`t make it either.
Only the horseshoe crab, one of the oldest living animals, older than dinosaurs, so old it is dubbed a "living fossil," produces this magic. And only in recent decades have scientists begun to fathom its power.
Chincoteague Island is a tiny, wind-swept place, more famous for its wild ponies than horseshoe crabs. It is here that crab-boat skipper Leon Rose brings his catch.
The BioWhittaker laboratory bleeds about 100,000 crabs a year, all of them caught by Rose. The process is fast, and lab officials say, painless.
The crabs are hosed down and sorted by size: The largest are about 2 feet long and weigh 10 pounds. The bleeding lasts a few minutes. Drained of about one-third of their blood, the creatures are tagged and tossed back into crates. Most survive.
That night they will be collected by Rose for their journey back to the deep.
After the bleeding, the science begins. The sapphire-blue blood is centrifuged to separate white cells from plasma. The white cells are then ruptured to release a protein, which is mixed with other ingredients and made into a freeze-dried powder called Limulus Amebocyte Lysate. LAL, as it is known, is sold to pharmaceutical companies around the world.
LAL was discovered in the early 1950s and became widely used in the 1970s. Before then, drugs were tested for contaminants by injecting a sample into a rabbit. If the rabbit got a fever or died, the drug was discarded.
Today, LAL is the most effective way to screen for contaminants in drugs and medical devices. Since 1987 it has been the required FDA test for all drugs used by humans.
The test is simple. The powdered LAL is rehydrated and a sample of a new batch of drugs dropped into the solution. If the drug is contaminated, the mixture will clot instantly.
Science hasn`t figured out how to make LAL synthetically.
The industry bleeds about 300,000 crabs a year, producing about $50 million worth of LAL. BioWhittaker bleeds between 600 and 1,000 crabs a day during the bleeding season, which lasts from about April to October. It makes about 600 liters of LAL a year.
Every spring, when the tide is high and the moon is full, millions of the ancient creatures crawl ponderously from the deep. They blanket East Coast beaches with their pearly green eggs. Then they creep back to the waves.
The birds swoop down, swarms of them: red knots, sanderlings, ruddy turnstones, gorging themselves on eggs to fuel their journey North.
The commercial fishermen sweep in, cleaning the beaches of thousands of crabs in a few hours. Female crabs are particularly prized as bait for conch and eel. The tourists come with their cameras. The researchers come with their notepads.
Suddenly, a mating ritual that hasn`t changed in 400 million years is being threatened.
Earlier this year, most Atlantic coastal states restricted by 25 percent the numbers that can be caught. The National Marine Fisheries Service declared a moratorium on commercial horseshoe-crab fishing in Virginia when that state refused to reduce its quota from 710,000 crabs to 152,495. In August, the U.S. government proposed turning the Delaware Bay into a national preserve.
Environmentalists say over-harvesting of the crabs has endangered rare migratory shorebirds whose survival depends on a Delaware Bay stopover. They also worry about the effect on the crab population.
In Japan, once another prime spawning area, overfishing has virtually wiped them out.
But fishermen argue the crabs are vital to the local bait business and that there are no indications of a serious decline.
The biomedical companies watch the debate anxiously, worried not just about the supply of blood. The horseshoe crab`s eyes are used for research on human eyes. The chitin from their shells is used for sutures.
"We are talking about the survival of a species that is ecologically, economically and medically vital," says Jim Berkson, assistant professor in the Department of Fisheries and Wildlife Sciences at Virginia Tech. "And no one really knows what is going on."
Posted Sep 9 2000 5:20PM
7 Echinoderms ...any of a variety of invertebrate marine animals belonging to the phylum Echinodermata, characterized by a hard, spiny covering or skin.
More than 21 classes have been identified, but only about 6,000 species of six classes are known to still exist. The six are:
Crinoidea ( feather stars and sea lilies),
Asteroidea (starfishes),
Ophiuroidea ( brittle stars and basket stars),
Echinoidea ( sea urchins, cake urchins, and heart urchins),
Concentricycloidea (sea daisies), and
Holothurioidea (sea cucumbers). Echinoderms are widely distributed in all the oceans, occurring in marine environments ranging from the intertidal zone to the deepest oceanic trenches.
The most apparent characteristics of echinoderms are the calcite skeleton and the five-rayed, or pentamerous, radial symmetry of the adult body form. The skeletal structure may be a test, a hollow structure formed of solid plates, as found in sea urchins, or it may be composed of many separate smooth or spiny ossicles held together by muscles or ligaments, which is the case in the starfishes.
Most echinoderms have numerous small appendages, called tube feet, that are contained in grooves on the animals' tentacles. Movement of the tube feet is controlled by a hydraulic, or water-vascular, system. Water is taken in and passes into five major canals that channel it into branches that carry it to the tube feet. Local muscle action in contracting or releasing the canals functions to extend or retract the tube feet. The tube feet themselves may be variously modified to serve in locomotion, respiration, tunneling, sensory perception, feeding, and grasping.
Reproduction among echinoderms is either sexual or asexual. In sexual reproduction, eggs are fertilized in the water by sperm and either pass through a larval stage before transforming into juvenile echinoderms or develop directly into juveniles. One kind of indirect development involves fission of the body and regeneration of the missing parts. Starfishes, for example, can regrow an entire organism from one arm if a small portion of the central disk remains attached.
Most echinoderms feed on microscopic detritus or suspended matter (either alive or dead), but many urchins and starfishes graze on plant life. Some starfishes are carnivores and eat mainly mollusks. A few of the echinoderm species are economically important. Some sea urchin roes and the warty sea cucumbers of the genus Holothuria are used for food in certain countries (see bêche-de-mer). Various species of starfishes, such as Asterias vulgaris and A. forbesi, that prey on clams and oysters are pests in commercial shellfish beds.
In most species the sexes are separate; i.e., there are males and females. Although reproduction is usually sexual, involving fertilization of eggs by spermatozoa, several species of sea cucumbers, starfishes, and brittle stars can also reproduce asexually.
Ecology. Habitats.
Echinoderms are exclusively marine animals, with only a few species tolerating even brackish water. Among the exceptions are a few tropical holothurians that can withstand partial drying if stranded on a beach by a receding tide. Most echinoderms cannot tolerate marked changes in salinity, temperature, and light intensity and tend to move away from areas where these factors are not optimal. The behaviour of a large proportion of shallow-water species is regulated by light; i.e., individuals remain concealed during the day and emerge from concealment at night for active feeding. Echinoderms are found in the warmest and coldest of the world's seas; those species that can tolerate a broad temperature range usually also have a broad geographic range. The horizontal or vertical distribution of many species is also governed by water temperature. The influence of pressure upon echinoderms has not yet been thoroughly investigated.
Echinoderms occupy a variety of habitats. Along a rocky shore, starfishes and sea urchins may cling to rocks beneath which sea cucumbers and brittle stars are concealed. Some sea urchins have special adaptations for coping with surf pounding against rocks (e.g., particularly strong skeletons and well-developed tube feet for attachment). In sandy areas starfishes, brittle stars, irregular sea urchins, and sea cucumbers may bury themselves or move on the surface. Large populations of all living groups of echinoderms can be found in mud and ooze offshore. In some marine areas, echinoderms are the dominant organism; in the deepest ocean trenches, for example, holothurians may constitute more than 90 percent by weight of the living organisms. Perhaps the most unusual habitat is exploited by sea daisies and a small family of asteroids; these animals occur only on pieces of waterlogged wood on the deep-sea floor.
Echinoderms frequently use other animals as homes; thousands of brittle stars, for example, may live in some tropical sponges. Sea cucumbers may attach themselves to the spines of sluggish Antarctic echinoids, and one sea cucumber attaches itself to the skin of a deep-sea fish. On the other hand, echinoderms are also hosts to a wide variety of organisms. Various crustaceans and barnacles, for example, cause the formation of galls, or tumourlike growths, in the skeletons of sea urchins, and crinoids are hosts of specialized parasitic worms. Commensal worms, which do no damage, are associated with most groups; an interesting case of commensalism is the association between various tropical sea cucumbers and the slender pearlfish, which often is found in the rectum of the holothurian, head protruding through its anus. Pinnotherid crabs may be found in the rectum of echinoids and holothurians in Peru and Chile, and highly modified parasitic gastropod mollusks are frequently found in the body cavities of holothurians. A conspicuous parasitic sponge grows on two species of Antarctic ophiuroids. Predation and defense.
Although echinoderm populations do not generally suffer from heavy predation by other animals, ophiuroids form a significant part of the diet of various fishes and some asteroids. Echinoids are frequently eaten by sharks, bony fishes, spider crabs, and gastropod mollusks; crows, herring gulls, and eider ducks may either peck their tests (internal skeletons) or drop them repeatedly until they break; and mammals, including the Arctic fox, sea otters, and humans, eat them in considerable numbers. Asteroids are eaten by other asteroids, mollusks, and crustaceans. Some holothurians are eaten by fishes and by humans. Crinoids appear to have no consistent predators.
Echinoderms can protect themselves from predation in a variety of ways, most of which are passive. The presence of a firm skeleton often deters predators; echinoids, for example, have a formidable array of spines and, in some cases, highly poisonous stinging pincerlike organs (pedicellariae), some of which may cause intense pain and fever in humans. Some asteroids use chemical secretions to stimulate violent escape responses in other animals, particularly predatory mollusks. Some holothurians eject from the anus a sticky mass of white threads, known as cuvierian tubules, which may entangle or distract predators; others produce holothurin, a toxin lethal to many would-be predators.
bêche-de-mer, plural BÊCHE-DE-MER, or BÊCHES-DE-MER, also called TREPANG, boiled, dried, and smoked flesh of sea cucumbers (phylum Echinodermata) used to make soups. Most bêche-de-mer comes from the southwestern Pacific, where the animals (any of a dozen species of the genera Holothuria, Stichopus, and Thelonota) are obtained on coral reefs. Bêche-de-mer is consumed chiefly in China. Bêche-de-mer, or Beach-la-Mar, is a pidgin English term used in New Guinea and nearby islands, where the trepang trade has long been important. The term Bêche-de-Mer has also come to designate the pidgin English language spoken in these regions.
Sea cucumber, any of the 1,100 species of marine invertebrates constituting the class Holothuroidea (or Holothurioidea) of the phylum Echinodermata. The soft, cylindrical body, 2 to 200 cm (about 3/4 inch to 6 1/2 feet) long and 1 to 20 cm (0.4-8 inches) thick, usually is a dull, dark colour and often warty, thus resembling a cucumber. The internal skeleton is reduced to numerous distinctively shaped, tiny bits in the skin. Most species have five rows of tube feet extending from mouth to anus. The anal opening is used for both respiration and discharging wastes. The 10 or more retractile tentacles surrounding the mouth are used for food-taking (mud containing nutrients or small aquatic animals) or burrowing. Locomotion is sluglike. Many sea cucumbers can expel their internal organs from the anus and grow new ones; this may be a device for escape from a predator, or it may occur for physiological reasons. Some species also expel sticky filaments that ensnare or confuse an enemy. Cucumbers shelter pearlfish (Carapus species) in the anal cavity, with the head of the fish extruding. A number of sea cucumbers exude a toxin that is lethal to small animals but not to humans; South Sea islanders place sea cucumber juices in water to kill or stupefy fish.
Sea cucumbers are found in all oceans, mostly in shallow water but sometimes at great depths. They are best represented in the Indian Ocean and the western Pacific. The 80 to 100 species of large, warty cucumbers of the genus Holothuria are abundant on coral reefs.
Sea urchin.... any of about 700 living species of echinoid marine invertebrates (phylum Echinodermata) with a globular body and a radial arrangement of organs, shown by five bands of pores running from mouth to anus over the test (internal skeleton). The pores accommodate tube feet, which are slender, extensible, and often sucker-tipped. From nodules on the test arise long, movable spines and pedicellariae (pincerlike organs); these structures may have poison glands. The mouth, on the underside of the body, has a complex dental apparatus called Aristotle's lantern, which also may be venomous. Teeth are extruded to scrape algae and other food from rocks, and some urchins can excavate hiding places in coral or rock--even in steel. Sea urchins live on the ocean floor, usually on hard surfaces, and use the tube feet or spines to move about.
The largest urchin (known from a single specimen) is Sperostoma giganteum of deep waters off Japan. Hatpin urchins, such as Centrostephanus longispinus of the Mediterranean and eastern Atlantic, Diadema (formerly Centrechinus) setosum of the Indo-Pacific, and D. antillarum of Florida and the West Indies, have toxic spines up to 30 centimetres (12 inches) long. The slate-pencil urchin (Heterocentrotus mammillatus) of the Indo-Pacific has 12-cm spines that may be 1 cm thick--stout enough to be used for writing. Lytechinus variegatus, a pale-greenish urchin of the southeastern coast of the United States and the Caribbean, and the large, short-spined Psammechinus (sometimes Echinus) miliaris of Iceland, Europe, and western Africa use their tube feet to hold up bits of seaweed or shell as a shield against sunlight in shallow water.
The small, reddish or purplish urchins of the genus Arbacia, such as A. punctulata, the common urchin from Cape Cod to the West Indies, are familiar subjects in embryology; a female may release several million eggs at a time. In the West Indies, sea eggs--the ovaries of Tripneustes ventricosus--are eaten raw or fried; in the Mediterranean region, frutta di mare is the egg mass of Paracentrotus lividus (the best known rock borer) and other Paracentrotus species; and, on the U.S. Pacific coast, the eggs of the giant purple (or red) urchin (Strongylocentrotus franciscanus) are similarly considered a delicacy. The slightly smaller S. purpuratus, of the same region, is known to excavate holes in steel pilings.
Cake Urchins... any of the echinoid marine invertebrates of the order Clypeastroida (phylum Echinodermata), in which the body is flattened. The surface is covered with short spines (often furlike) and inconspicuous pedicellariae (pincerlike organs). In many species the hollow, slightly elongated test (internal skeleton), which accommodates the water-vascular system, is symmetrically notched on the edge or has narrow perforations (lunules). The upper surface of the test has a radial, flowerlike arrangement of five porous spaces, called petaloids; the pores permit the extension of tube feet modified for respiration. Tube feet on the underside of the body are used for taking food and may also assist the spines in locomotion.
Found worldwide in shallow seas, cake urchins burrow in sand and feed on organic particles wafted to the mouth on water currents. They are in turn the prey of other echinoderms and mollusks.
Cake urchins in which the body is a coinlike, thin-edged disk are called sand dollars, a name sometimes given to cake urchins generally. The test of the common sand dollar (Echinarachnius parma) is often washed up on beaches of North America and Japan. Species with lunules are generally called keyhole urchins. The largest and thinnest cake urchin is the yellow or purple sea pancake (Echinodiscus auritus) of the East African coast.
Sand dollar, any of the invertebrate marine animals of the order Clypeastroida (class Echinoidea, phylum Echinodermata) that has a flat, disk-shaped body. It is a species of cake urchin, intermediate between sea urchins and heart urchins. The sand dollar is particularly well adapted for burrowing in sandy substrates. Very small spines used for digging and crawling cover the entire surface of its body and are appressed backwards toward the posterior anus. The mouth is located in the centre of the body's underside. The upper surface exhibits pentaradiate symmetry, with a pattern of five "petals" spreading out from the centre. Some species found stranded on the shores of North America have five or six slots, or lunules, through the test (external skeleton). Most sand dollars measure from 5 to 10 cm (2 to 4 inches) in diameter. Species of comparable size occur in shallow coastal waters throughout the rest of the world, except in Europe and Antarctica.
Heart urchin, any echinoid marine invertebrate of the order Spatangoidea (phylum Echinodermata), in which the body is usually oval or heart-shaped. The test (internal skeleton) is rather fragile with four porous spaces, or petaloids. The body is covered with fine, usually short spines. Heart urchins live in burrows lined with mucus. Long tentacles (modified tube feet) reach out over the sand to pick up small particles of food; other tube feet have respiratory and sensory (not locomotive) functions. Movement is carried out by means of the spines. The common heart urchin (Echinocardium cordatum) occurs in all oceans. Spatangus purpureus is common on the coasts of western Europe, the Mediterranean, and western Africa.
8. About Lobsters.... SPINY LOBSTERS, CALLED 'FLORIDA' LOBSTERS
They are not American lobsters -- which are called 'Maine' lobsters. If you can't tell them apart by looking at them, you could always ask their names: Homarus americanus with two big meat-filled claws and Panulirus argus with two ordinary front legs where the claws would be. The spiny lobster, lacking claws, has two long, thorny antennae which it uses as whips for fighting and defense. Both kinds of lobsters are jointed-leg animals like their crustacean relatives the crabs and shrimps. But spiny lobsters are distinct in more ways then in name and claws. There are many species, six in the western Atlantic (P. argus being the most common), with small anatomical differences but large variations in color, ranging from dark brown to red, yellow and even blue, and mottled for camouflage. They are found worldwide in tropical and temperate waters, most notably off Australia and South Africa, both of which are big lobster exporters. Needless to say, wherever found they are very tasty, the main reason they have been overfished. Spiny lobsters start life in a cluster of thousands of eggs beneath the tail of the female. There they ripen until one day...she sheds them into the open sea where they begin a perilous odyssey in the plankton, molting eleven times before finally changing into a shape that even the untrained eye can recognize as 'lobster'. A few tiny survivors, which may have been spawned hundreds of miles away, are carried inshore by currents and settle in sea grass beds and around the roots of the red mangroves where they feed on both animal and vegetable matter. When about three inches long they again migrate to deeper water. There under ledges and in holes they hide by day and feed by night, mostly on carrion, small snails and clams. In response to the season or food availability, they move en masse, often in columns hundreds of yards long, from shallower to deeper water or along the shore. They grow by periodically molting. They cast off their old shell, swell body tissues with seawater and await the protective hardening of the new shell into which they will flesh out. Growth slows as they age and molting becomes infrequent. Still, a spiny lobster in Gulf waters can attain a length of 20 inches and weigh over 10 pounds. Other species in other parts of the world grow even larger. Spiny lobsters can crawl forward and sideways but always swim backward with flaps of their powerful tail, a gesture that presents an armored, thorny face to a potential enemy. Those include octopuses, turtles, nurse sharks and rays none of which eat them with melted butter, a la Newberg or cold with mayonnaise like their greatest enemy, man. During the spawning season spiny lobsters are protected by law.
SAND-BURROWING Amphipods-- A North American Success Story by John Foster V17#3 UN
The amphipod crustaceans variously called sand-burrowers, beach fleas, or sand-hoppers represent several families. They all occupy the littoral zone, either burrowed in the sediment or nestling in the strand line among debris. It is the burrowing variety, the family Haustoriidae, which is seldom seen. A casual observer may pick up a clump of algae or displace a washed-up piece of wood and it may explode with jumping arnphipods, probably Orchestia or Talorclrestia, but a beachcomber will require a scoop and a screen to see the haustoriids, or sand- burrowers.
These interesting and well adapted crustaceans are found in sandy environments both on wave-pounded beaches and in quiet inlets and channels. People who stroll the sandy beaches of the Atlantic and Gulf coasts will be curious about the role, behavior, and diversity of these obscure crustaceans.
The haustoriids are small to medium sized arnphipods, usually one to ten millimeters long. The family gets its name from the Latin word meaning "drawer of water." The significance of their name will become clear as their feeding and burrowing habits are considered. Within the family, there are variations in form. One subfamily, comprised of more slender forms such as Ampliporeia are equipped with mouthparts best adapted to "sand licking," that is, grazing upon the organic coating of the sand grains which compose the sediment. In Amplriporeia, and in similar American genera like Pontnporeia and Barthyporeia, the first segment of the first antenna is pronounced in such n way as to give the appearance of a prow. The members of this subfamily also show physical differences between males and females. This is known as sexual dimorphism.
The focus of this article is the other side of the family, the subfamily known as Haustoriinae. It is composed of a more specialized group of amphipods with broad bodies and strong appendages for burrowing. Richly setose and spiny mouthparts, adapted for filtering interstitial water for food particles, are also characteristic. The general body architecture creates what amounts to a three-sided tunnel. The body trunk, known as the pereon, is made up of seven segments. It forms the top of the tunnel. Connected to the pereon and projecting downward are seven pereopods, or legs, with broadened, plate-like segments. They form the sides of the tunnel. The next three segments, known as the pleon, are equipped with a pair of pleopods, or swimmerets, per segment. The movement of the pleopods establishes a forward directed current of water over the gills (attached to the top, inner surface of the legs) and toward the mouthparts. An opposite, or posteriorly directed current, may be set up by movement of the broad lobes of the antennae. The ultimate destination of the food-bearing water is the mouthframe, comprised of a pair of mandibles, two pairs of maxillae, and a pair of maxillipeds. The food particles are trapped on the setae and spines of the various parts and transferred to the mouth.
While most of the haustoriins look superficially similar, small differences in the form and function of the appendages, especially the mouthparts, separate them into genera and species. There is a distinct relationship between the structures of a genera or species and its place in the habitat. Before examining some of the various forms, a review of the difficult habitat of these crustaceans will be useful.
Animals dwelling in the sandy beach habitat require a high degree of adaptation for survival. The environment is usually unstable, depending on the location and wave-energy. It is usually highly oxygenated and comparatively silt free. Amphipod inhabitants have to cope with, in addition to the violence of wave stress, the grinding, abrasive action of moving sand, potentially rapid changes in temperature, salinity, and exposure, and the absence of any solid substrate around which to seek refuge. On the plus side, in addition to the presence of oxygen, there is a large food supply which is constantly replenished by rising tides and wave wash. Further positive points can be made for the intertidal sand habitat. While to us it seems uniform and sometimes barren and hostile, to marine life it offers a wide variety of ecological niches with enough diversity to allow several sympatric species (those living in the same habitat) to coexist. The reverse beauty of the situation is that the various haustoriins occupying the habitat, although structurally similar, are sufficiently diverse to partition the environment to meet the needs of their own niche.
Evidence for evolutionary changes in the sand-burrowers is found initially in their truncate, broadly fusiform bodies, widened side plates, and powerful pereopods and pleopods for burrowing and pumping water. The most significant adaptation in structure which allows them to divide the resources of the habitat, and coexist, is found in the mouthparts. Differences in mouthparts reflect differences in ecological station. Whether a group is intertidal or subtidal (aquatic) depends, to a large degree, to the adaptation of their mouthparts for filter feeding.
Sand-burrowing amphipods may be generally divided into intertidal and subtidal forms. The haustoriins with the least modification of their mouthparts for filter feeding are subtidal. Examples are the genera Pseudohaustorius and Protohausorius. These, and others which occupy the subtidal zone exclusively, may be
found along high energy, wave-exposed beaches or shelly to muddy, partially sheltered flats, from the lower intertidal zone to depth of six or seven meters. They feed on organic materials extracted from the water along with microorganisms and small invertebrates. Both genera listed above are considered primitive, that is, they arc closest to the stock from which all other North American haustoriin genera have radiated.
A much more specialized group, using a habitat not suitable to less adapted groups, is comprised of the intertidal forms such as Neohaustorius, Haustorius, and L.epidactylus. They use the interstitial water to its maximum potential. The intertidal forms occupy the coarsest and least silty sands. They feed quite efficiently on the organic food in the water which is constantly renewed through tidal and wave activity. Neohaustorius is a genus which has not been collected to date in the Gulf of Mexico. The best known member of this genus is Neohaustorius schmiizi which has been studied extensively in North Carolina. This species does not attain the population densities of its relative Haustorius. It is normally found in densities about 100 individuals per square meter and occurs from Canada to the Atlantic coast of North Florida. Neohaustorius schmitzi feeds on detritus and the bacterial film on the sand grains. Gut analyses have revealed small amounts of fine sand and diatoms in these amphipods. The diet of fine sand allows this species to obtain adequate nutrition because of the higher level of bacterial growth present. Haustorius, on the other hand, which occupies the same habitat, includes minute interstitial invertebrates such as polychaete worms, nematodes, and copepods in its diet. It does so because its particular niche is found in coarser sands with generally less bacterial growth. Both species are high tide feeders. Croker, who did much of the early work on the diets of haustoriids, observed that within two to four hours after the sand was exposed by low tides, most animals examined had empty guts.
Lepidactylus appears to prefer quieter beaches. It is found in inlets, bays, and estuaries. In the Gulf of Mexico, this genus is very abundant in summer, at times occurring in densities of 1500 individuals per square meter of sediment. A genus which also occurs in normally quiet, sheltered waters, in habitats similar
to that of Lepidactylus, is Acanthohaustorius. It is so named because of the strong spine located on the last segment of the pleosome. It occurs, as at least eight separate species, from Maine to Texas in the midtide zone to shallow water. The greatest concentrations are found about the mean low water level. In St. Andrew Bay, Florida, this genus is found on the sandy bayshore, sometimes in areas vegetated with seagrasses . In winter, when bay water temperatures drop into the 50°F range, Acanrhohaustorius is easily collected in the wet sand just at the low tide line or in very shallow water. In summer, when the surface water temperatures arc in the 80°F range, one must collect in waters one to four meters deep in order to locate any sizeable numbers. Acanrhohaustorius apparently migrates with the seasons, at least in St. Andrew Bay. Other members of the genus have been collected in deep waters (40 meters) off Long Island, in shell hash sands off the east coast of Florida, and in coarse coralline sands in the Florida Keys.
How can these various amphipods, which appear to use the same resources, occur sympatrically in the sandy beach habitat. The answer is complex, but to simplify, it can be said that they each occupy a distinctive niche. An ecological niche is, simply stated, a particular combination of physical habitat characteristics, community roles, and behaviors which make the species distinctive within the ecological community. When similar species occur together, it must be assumed that there is a zone of non-overlap in their individual niches. There arc two properties which allow this coexistence. First, mechanisms must exist which insure reproductive isolation. These may include spatial separation, habitat isolation, seasonal breeding isolation, and barriers to cross fertilzation. Secondly, the ability must exist to use the same resource as another species in a distinctive way. The haustoriids described above have accomplished this adaptation, in part, through staggered breeding seasons. The subtidal species tend to breed in late winter and early spring while the intertidal species breed in late spring and early summer. In this formula, the young do not have to compete for food and space. Additionally, as we have seen, the haustoriins have divided the habitat by using different food resources and feeding strategies.
The success of American haustoriins is borne out by the fact that the North American Atlantic coast possesses most of the known genera and the greatest number of endemic genera and species of the subfamily Haustoriinac. Dr. E. L. Bousfield of Canada, a world authority on haustoriids, has pointed out in a 1970 paper on the evolution of the group, that the ability of the subfamily Haustoriinae to inhabit more landward and more estuarine areas relates to the increasing specialization and efficiency of its filter feeding apparatus and its reproductive cycle, these features limit competition among the young of the sympatric family members, a process known as adaptive radiation. It occurs when the offspring of some common ancestor, which has moved into a new environment, undergoes rapid evolutionary changes in the form and function of the body necessary to meet the demands of the new environment.
The sand-burrowers, while perhaps mundane to some, provide us with art evolutionary success story. The descendants of the ancestral stock haustoriin, thought to be closely related to Protohaurstorius, have become altered and better adapted to the process of expanding geographically. This expansion, which has been from the North Atlantic to the Gulf of Mexico, also includes the opening of new habitats, from wave-pounded outer beaches to quiet, estuarine bayshores. Natural history has many glaring examples of short-lived, but flamboyant animal groups without the resilience of these adaptable, obscure sand-dwellers.
9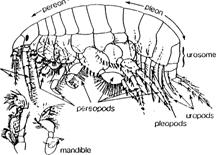
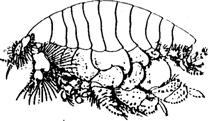
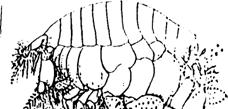
Haustorius Acanrhohausrorious
Name____________________________________pd_____
1. Sea Moss
1. What is the main idea of the “Article”?
2. What scientific principle(s) are illustrated in the “article”?
a) ____________________________________________________
b) ____________________________________________________
c) ____________________________________________________
3. What are 5 facts stated in the “article”?
a) _____________________________________________________
b) _____________________________________________________
c) _____________________________________________________
d)______________________________________________________
e)______________________________________________________
4. What is the conclusion of the “article”?
2. Tube Worms
1. Where was the giant tube worm found?
2. How do pogonophorans feed?
3. Where do bacteria get their energy?
4. How are shrimp around the vents different than the normal shrimp near the surface?
5. How hot is the water running out the vents? _______What about water about a foot away from the vent?
6. What is the most prevalent chemical dissolved in vent water?
7. How many bacteria are in an ounce of tissue?
8. How do tube worms reproduce?
9. What and where is Godzilla?
10. What happens when the vents stop flowing?
3. Brachiopods Ancient Dwellers of the Sea
1. Describe the lopophore and explain its functions.
2. Where would you find the living brachiopods
3. Compare and contrast the 2 types of Brachiopods
.Articulates | Inarticulates
4. Spiders
1. What is the main idea of the “Article”?
2. What scientific principle(s) are illustrated in the “article”?
a) ____________________________________________________
b) ____________________________________________________
c) ____________________________________________________
3. What are 5 facts stated in the “article”?
a) _____________________________________________________
b) _____________________________________________________
c) _____________________________________________________
d)______________________________________________________
e)______________________________________________________
4. What is the conclusion of the “article”?
5. Respect for Old Timer QUESTIONS FOR HORSESHOE CRAB
1. Is the horseshoe crab as dangerous as it looks?
2. What is the Genus and species of the horseshoe crab?
3. Why do humans destroy horseshoe crabs?
4. What creature is the horseshoe crab most closely related to?
5. How long have they been around on earth?
6. Where, geographically, are horseshoe crabs found and to what depth would you find them?
7. When is the breeding season?
8. Why are people afraid of the horseshoe crabs?
9. Which is larger, the male or female crab?
10. How long do the eggs take to hatch and where do they hatch?
11. Beside the size, how do young differ from adults?
12. What is the function of the telson?
13. Where is the mouth located?
14. How does the horsechoe crab breath?
15. Which part of the horseshoe crab is edible?
16. What were these crabs used for by man in the past and what are they used for now?
17. What would you have to do to keep one successfully in a marine tank?
18. What is todays cause of the decline of the horseshoe crab?
19. Where did this article come from?
20. Have you ever touched a horseshoe crab?
6. BlueBloods in Danger
1. What is the main idea of the “Article”?
2. What scientific principle(s) are illustrated in the “article”?
a) ____________________________________________________
b) ____________________________________________________
c) ____________________________________________________
3. What 5 facts stated in the “article”?
a) _____________________________________________________
b) _____________________________________________________
c) _____________________________________________________
d)______________________________________________________
e)______________________________________________________
4. What is the conclusion of the “article”?
7. Echinoderm reading
1. What is trepang and how is it used?
2. How do sea cucumbers get their name?
3. What do south islanders use sea cucumbers for?
4. Which organs may have poison glandson the sea urchin?
5. What is Aristotles Lantern and how is it used?
6. Which species of sea urchins have 12” long toxic spines and where are they found?
7. Why do sea urchins hold up bits of seaweed ?
8. What part of the sea urchin is eaten?
9. What are petaloids and what is its function?
10. What is he difference between the habitat of sand dollars and heart urchins?
11. How many classes of Echinoderms are known and how many are around today?
12. Describe both types of reproduction in echinoderms.
13. At what water temperature do echinoderms prefer?
14. In what type of habitats are echinoderms found?
15. What are usual predators of Crinoids?
9. Sand-Burrowing Amphipods
1. Where are amphipods found?
2. Where would one find haustoriidi?
3. How large are the haustoiids?
4. What is “sand lickng”?
5. Describe the pereon of sub-family Haustoriinae?
6. How do the pleopods function?
7. Why are amphipod inhabitants of sandy beaches?
8. How is the food supply replinished?
9. What is meant by “sympatric species”?
10. Why would the mouth parts determine the ecology of these animals?
11. On what does Neohaustorius schmitzi feed?
12. How are breeding seasons different in subtidal and intertidal species?
13. What is an ecological niche?
14. What did the increase in the efficiency filter feeding and reproduction of Haustoriinae bring about?Sand-