Canaveral
National Seashore serves as an important nesting area for sea turtles. During
the months of May through August, giant sea turtles lumber ashore to nest on
the beach. Three species are known to nest within the park: the loggerhead,
leatherback and green sea turtles. Loggerheads lay 3,000-4,000 nests per year.
Up to three hundred greens and only a few leatherbacks deposit their eggs
within the park boundaries each nesting season.
The
sea turtles lay approximately 100 round, white, leathery eggs in each nest.
Prior to 1984, most of the eggs laid
within the seashore were eaten by raccoons, and to a lesser extent by ghost
crabs. Some nests are lost when beaches erode during storms.
If
the eggs survive, they begin to hatch in about sixty days. The first turtles to
hatch will wait until their nest-mates have left their eggshells. Because of
the depth of the nest, it would be difficult for one three-inch hatchling to
emerge from the eighteen-inch deep nest by itself. There is also safety in
numbers. When the cool sand signals the safety of nighttime, the hatchlings
gradually dig their way out of the nest in a united effort to make their way to
the sand’s surface.
Many
hazards await the hatchlings when they reach the surface of the nest. Ghost
crabs, birds, raccoons and the drying heat of the early morning sun are waiting
for the tiny turtles as they try to make it to the ocean.
Once
the turtles make it past the surf, they swim to a region of the Atlantic Ocean
known as the
The
ocean holds more hazards than just the sea turtles’ natural predators. Many
deaths are attributed to entanglement in fishing lines, collision with ships
and boat propellers, drowning in commercial fishing nets and ingesting plastic
fragments or congealed oil.
Research on the Park
In the early 1980’s, researchers found raccoons
were destroying 98 percent of Canaveral’s turtle nests. The park began a
program to help reverse this trend. After experimenting with several different
ways of protecting the eggs, park rangers found that by securing a wire mesh
screen over the nest, raccoons were prevented from digging into the nests. The
small hatchlings could still exit the nest through the openings in the mesh.
During the months of May through August, park rangers and volunteers work each
night to screen the nests. After two months, when all the eggs have hatched,
the screens are removed and one of every ten nests IS excavated to monitor the
success rate. Over 80 percent of the turtle nests have been protected using
this method.
YOU Can Help
1. Possessing turtle parts is illegal. Leave any
dead sea turtle you may find undisturbed. Write down where you found the turtle
and, if it was tagged, the number, color and type of tag. All sea turtles are
classified as either threatened or endangered species, and are protected by
federal law. Moving a carcass or removing pa~ts from one is prohibited by the
Endangered Species Act. If the turtle was found in Canaveral National Seashore
report the finding to the
2. Canaveral National
Seashore is closed at night, but should you encounter turtles on a beach
outside the park, turn off flashlights and car lights and do not interfere with
the turtle’s activities. If a sea turtle is disturbed, it will not nest.
3. Attend a conducted
program or a nighttime Turtle Watch” to learn more about sea turtles. Turtle
Watches take place at night in June and July. Ask a park ranger for details.
1.
THE GREEN TURTLE
by BLAIR E.
WITHERINGTON
V18#1 1989
Underwater Naturalist
Witherington is a doctoral candidate in
zoology at the
Few reptiles have influenced man’s culture,
inspired his curiosity, and roused his admiration more than the green turtle.
Though this turtle’s beauty and mysterious nature may prompt romantic prose,
it is the utility of its body parts which has given it the dubious distinction
of being the world’s most important reptile.
The use of the green turtle as food has
both cultural and historic implications. European sailors first exploring the
In modern times, the green turtle and its
eggs continue to be valuable sources of protein for many coastal subsistence
cultures. In many cases, centuries of tradition surround the capture and use
of these animals. Apart from their nutritive value, green turtles have also
played a part in folklore, touted as sources for various medicines and
aphrodisiacs.
Many developed countries tend to view the
green turtle primarily as a source of valuable luxury products. Perhaps the
most renowned of which is the clear broth made from the fatty cartilaginous
tissue attached to the animal’s upper and lower shell. These tissues,
individually called calipee and calipash, are typically surrounded by a green
fat from which the turtle takes its name. Other common names such as “soup
turtle” and “edible turtle” reflect the special culinary regard held for the
green turtle. Cosmetic oils, fine leather products, and stuffed whole animals
complete the list of luxury products.
The green turtle is a splendidly handsome
animal. The head is smoother and more rounded than in other sea turtles. Its
carapace is oval-shaped with a slight posterior tapering, moderately domed, and
covered with smooth non-overlapping scutes or plates. Hatchlings are roughly
two inches in shell length and weigh about one ounce. The soft carapace and
most of the upper surface is typically a solid blue-black. Thin white margins
on the trailing edges of the front flippers and the cream white underside
contrast sharply.
The soft carapace hardens with growth and
develops a light, chocolate brown coloration that spreads from the center of
each carapace scute. By the time the animal is the size of a dinner plate, a
pattern of radiating streaks originating within each of these scutes can be
clearly seen. Shades of yellows, browns, greens, and black in the pattern vary
among individuals.
Green turtles are the largest of the
shelled sea turtles, growing to about three feet in carapace length and 250
pounds. Historical accounts report adult weights of over 600 pounds, but few
contemporary adults weigh much over 400 pounds.
Adult shell coloration varies both between
and within populations. The bright, radiating lines of the juvenile pattern may
remain or fade into a buff, grey, or olive tone, often with scattered dark
spots.
If the green turtle’s appearance is considered
dainty, then so might be its temperament. When captured, green turtles seldom
thrash wildly or attempt to bite. A few minutes after being boated, green
turtles are placid and submissive, unlike their loggerhead, hawksbill, and
ridley cousins. In the water green turtles are generally shy. offering divers
only fleeting glimpses.
The green turtle, Chelonia mydas. shares its genus, Chelonia, with a closely related turtle from the eastern Pacific,
the black turtle (Chelonia agassizi). Although
the species status of the black turtle may be the subject of taxonomic dispute,
the distinctness of its population is clear. Black turtles, as their name
suggests, are darkly pigmented on both top and bottom. The adults are
considerably smaller than green turtles, and they lay smaller complements of
eggs.
Green turtles are powerful, swift swimmers,
but despite their aquatic grace, they are, like all sea turtles, awkward on
land. Green turtles leave the sea to nest on sandy beaches. They move with a
sort of butterfly stroke gait using all four flippers simultaneously. This
gait differs markedly from the alternating limb movements typical of terrestrial
quadrupeds and most other sea turtles.
Most of what is known about green turtle
life history has been pieced together from information gathered on nesting
beaches when the turtles return to the terrestrial environment, dig nests,
deposit their eggs and conveniently place themselves within the reach of
scientists. But only a small portion of the turtle’s life is spent on a nesting
beach. Biologists wishing to conduct a complete study of green turtle ecology
are limited by the physical barriers that the vast oceans present.
For the most part, studies on the aquatic
lives of green turtles has taken a “black box” approach. One indirect yet
useful technique is a turtle-tagging program. Nesting green turtles are easily
tagged. The tags are commonly attached to the turtle at the trailing edges of
the front flippers and are engraved with an identifying code and the tagger’s
return address. Like messages in bottles, tags may be recovered later at the
same beach or elsewhere. Comparisons of information gathered from tagged and
recovered green turtles serve as clues to their oceanic biology.
On the eastern shore of
Recoveries of tags applied at Tortuguero
indicate that female green turtles travel hundreds of miles between nesting and
foraging areas. In addition to being extensive, these migrations also appear to
be incredibly precise. Green turtles make their landfalls often within yards of
the same sites they used in previous seasons. This site fidelity may greatly
restrict the genetic exchange between nesting colonies. Although many nesting
green turtles have been tagged throughout the
The incubation period of green turtle
clutches is roughly two months, depending on ambient temperature. When nearly
all the hatchlings in a clutch have emerged from their shells, the little
turtles scrape away at the surface of the egg chamber beneath the sand in an
orchestrated fashion. This sibling teamwork displaces nearly three feet of
sand while the hatchlings make their way up the column dug by their mother.
Under cool conditions, typically at night, the hatchlings waiting at the
surface of the nest burst from the sand in unison and scramble toward the
ocean.
Once in the surf they stroke away from
shore along relatively straight paths for miles beyond their sight of land.
This pelagic trek marks the beginning of the “lost years,” one of the most
enigmatic aspects of green turtle life history. Hatchlings leaving their natal
beaches are apparently incorporated into pelagic gyres
that may span entire oceans. Within these
gyres, colliding surface currents concentrate the small turtles within
floating communities of free living and epibiotic animals. The common marine
alga Sargassum forms the substrate
on which many of these communities depend. Many sea turtle biologists believe
that it is this collection of pelagic coelenterates, crustaceans, mollusks,
worms, and other invertebrates on which green turtles and other sea turtles
feed during their first few years. Unfortunately these same biological fronts
tend to collect oil, tar, and other synthetic refuse.
After spending these first few years as a
pelagic carnivore, green turtles become vegetarians. At four to eight pounds in
weight, juvenile green turtles move to the underwater seagrass pastures that
cover the shallows of continental shelves and island lagoons in the tropics
where they eat primarily seagrasses and benthic algae. From this point on they
remain benthic herbivores. Only manatees and dugongs are larger marine
herbivores. In captivity green turtles will feed readily on cut fish and squid.
Turtles on such high protein diets grow much faster than their wild counterparts,
but may suffer some liver dysfunction in later years.
Adaptations to their herbivorous lifestyle
include sharp serrations or “pseudo-teeth” outlining the edges of the lower
beak. These serrations slide against grooves in the upper jaw and act as efficient
grass clippers. By cropping rather than uprooting seagrasses, green turtles are
able to maintain regular feeding areas much like well-kept lawns. It appears
this continued cropping improves the nutrient quality of the seagrasses.
Tagging and recapture studies of juvenile green
turtle populations reveal that it takes 30 or more years for them to reach
sexual maturity. As green turtles approach adult size, they grow much more
slowly. Adult females measured repeatedly at nesting beaches over as many as 10
years often show scarcely more than half an inch
of growth in shell length. Because adult
green turtles grow so slowly, accurate judgments of adult age based on size are
impossible. Although some green turtles may in fact be quite ancient, it is probable
that most large individuals simply reached maturity at an especially large
size.
The International Union for Conservation
of Nature and Natural Resources (IUCN) regards the green turtle as an endangered
species. The historical record indicates that many of their unique nesting
colonies have been extirpated or have diminished to paltry numbers of individuals.
Most of the green turtle rookeries which once provided a seemingly endless
supply for the original Caribbean colonists have been virtually eliminated,
among these the Cayman Islands,
Although well deserved, the green turtle’s
endangered designation should not lead one to believe that this turtle’s numbers
are well known. The demographical data necessary to formulate population
dynamics models are difficult to obtain. Even if censuses of the world’s populations
were complete, it would be difficult to say for sure whether the numbers acquired
were adequate for population stability.
Fortunately, those interested in conserving
green turtles have not, in the
face of such uncertainty, been hesitant to
act on behalf of this species that appears to be slipping dangerously toward extinction.
Many nations now regulate or prohibit the taking of adult females on nesting
beaches. In some countries the harvest of and trade in green turtles from egg
to adult stages are banned. In addition to protection, governments are experimenting
with various other conservation programs ranging from shielding nests from
predators to rearing hatchlings until they are large enough to avoid predations.
Efforts to conserve green turtle populations
are not immediately rewarding. Because of the green turtle’s long generation
time, successful conservation practices initiated today will not result in appreciable
positive changes in numbers of nesting green turtles for decades to come.
Nesting populations in areas where lengthy protection has been afforded, such
as the
The
survival or extirpation of the world’s green turtle populations will depend on
the empathy and resolve of both developed and developing nations. The
preservation of this truly international and magnificent animal will prove to
be a challenging and worthwhile endeavor.
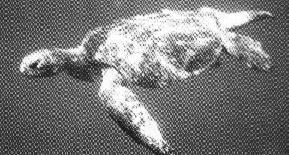
THE
LEATHERY TURTLE
by KAREN and SCOTT ECKERTThe authors received
doctorates from the
The
leatherback sea turtle, known to scientists as Dermochelys coriacea, is the
sole surviving species of the family Dermochelyidae. Arguably the most oceanadapted
of all the sea turtles, the leatherback boasts several unique morphological
features. Powerful front flippers, spanning 6.5-8 feet, are ideal for pelagic
existence where endurance to traverse vast expanses of open ocean is the key
to survival. In contrast, other sea turtle species, all members of the family
Cheloniidae, are best described as littoral in their ecology. They have bony
shells and their front flippers are proportionally shorter than those of the
leatherback, allowing chelonids the advantage of quick, acrobatic movement.
Skillful maneuvering is important in shallow water where foraging can entail
exploring the crevices of a reef or rock ledge, and where predator escape
requires an instant response.
The
most notable feature of the pelagic leatherback is its streamlined form. Broad
shoulders, a strongly tapered carapace, and seven dorso-longitudinal ridges
serve to enhance laminar flow. The leatherback is sheathed in leathery dermis
rather than a rigid bony shell. This characteristic gives the turtle its name
"dermo chelys," or "skin turtle." Streamlining and bone
reduction allow these 600-1200 pound giants to glide through the water with
minimal resistance. Popular names, such as "trunk,"
"coffin-back," and "keelback," reflect its unique body
form. The leatherback's uniqueness is more than skin deep, however. The
carapace, rather than being held rigid by fused ribs, is supported internally
by a mosaic of small bones linked together like a jigsaw puzzle and embedded
in tough, oil-saturated connective tissue. The ribs lie underneath this mosaic
and are not flared appreciably nor fused together. The plastron, or belly
plate, consists of eight narrow bones joined in a broad open ring and embedded
in thick dermal tissue. The somatic skeleton shows more cartilage than other
sea turtles and is theoretically more flexible. Clearly this streamlined,
smooth-skinned, flexible turtle is quite unlike its armored tank cousins.
All
these modifications make sense when one remembers what a leatherback does for a
living. Ranging the world's oceans in search of a bizzare diet of jellyfish,
there is little need for the protection that bony shell confers against
abrasion. Hydrodynamic form is much more important. In addition, internal
flexibility may be requisite for deep diving. We have recorded gravid
leatherbacks diving to depths exceeding 3200 feet in the
Leatherbacks
rarely, if ever, rest at sea. This incessant movement may be important for
survival in the wild, but it is a disappointment for scientists who would like
to keep leatherbacks in captivity. Husbandry techniques have yet to be
successful. Leatherbacks, even tiny hatchlings, swim against the walls of confinement
and often fatally injure themselves. In light of all that has been said, it is
not surprising to learn that leatherback nesting beaches are chosen as much
for their offshore characteristics as for their terrestrial amenities. Since
offshore reef and rock would abrade or seriously injure the turtles as they
attempted to land, favored nesting beaches, such as those in
Nesting
presents many challenges for sea turtles. Predation risk, elevated body
temperature, and fatigue characterize the terrestrial sojourn. Strong, slender
flippers confer freedom and grace in the ocean but are poorly suited for beach
crawling. Gravid leatherbacks use a heaving "breast-stroke" to
propel themselves forward at a labored pace of 10 feet per minute. Since they
lack reverse gear, a female nesting in French Guiana or eastern
that a
turtle returns as an adult to nest on the beach from which she herself was
hatched. In this way, those beaches most successful in producing baby sea
turtles will be favored by the next generation. Thus, over time, beaches poorly
suited for leatherbacks for reasons of high predation, dangerous surf or
confusing lagoons, are weeded out of the species' collective memory simply
because the turtles that nested there did not survive, or did not leave
surviving offspring.
Leatherbacks commonly nest every two or three years. The
nesting season is a lengthly one, spanning March to Julythroughout
most of the Caribbean and October to February along the Pacific coast of
We have no answers to this puzzling pattern of arrival, but it
is interesting, in this high-tech age, that one of the most useful tools
available to us may turn out to be that familiar little hitch-hiker, the
barnacle. These "living tags," picked up through successive
latitudes, offer clues about the general direction of migratory movement. Our
studies of barnacles on leatherbacks nesting at Sandy Point National Wildlife
Refuge,
Nesting
is typically nocturnal. Once ashore, a nest site is chosen, and the rear
flippers are used to excavate an egg chamber. During this process, one flipper
and then the other scoops the sand. As the flask-shaped cavity nears
completion, it is as deep as the flippers can reach. Since these flippers are
strongly modified for ruddering in the open sea, one cannot help but marvel at
how well they perform their adopted task. As the flipper reaches slowly,
deliberately, to the bottom of the nest for the next scoop of sand, it
articulates just as a human hand would do in the same position. It scoops, cups
the sand, and curls upward to avoid spilling the load. Each load is carefully
dumped at the nest rim, then expertly flung over the carapace to preclude its
sifting back into the nest. An experienced field biologist can tell at a glance
the stage of the nesting cycle by the pattern of discarded sand saddling the
female's carapace.
When the cavity is complete
(25 - 30 inches deep and 10 inches wide), the female rests with one rear
flipper poised over the nest. One imagines that this posturing evolved to
shield the eggs from rain and obscure them from waiting mongooses, coati
mundis, and other predators. Within seconds, the eggs drop into the nest. One,
then two, and three at a time. After all the preparation, the main event is
over in less than nine minutes. Fifty to 120 glistening, white eggs come to
rest gently against one another. These leathery, soft eggs are larger than
those of any other sea turtle. Their diameter is consistent across populations
and averages about two inches. The clutch also includes as many as 40 or 50
smaller, often misshapen, yolkless eggs. The function served by the yolkless
eggs is unknown. The leatherback is the only turtle that lays so many nonviable
eggs.
The eggs are
covered and left to incubate in the warmth of the sun. They are highly dependent
on favorable outside conditions for development. Temperature is one of several
crucial environmental parameters. It determines incubation duration, affects
moisture regimes and, when too high or too low, can cause deformity or death of
the tiny embryo. Perhaps the most interesting aspect of thermal ecology in sea
turtles is that the sex of the baby turtle will be determined predominantly by
the temperature at which the egg incubates. A shift of less than one degree
during the critical period (believed to be the middle trimester of the
two-month incubation) can bias sex ratios toward one sex or the other. Warmer
temperatures result in more female hatchlings, cooler temperatures in more
males. The pivotal temperature, that temperature at which the clutch contains
both males and females in roughly equal proportions, has been reported to fall
between 29 and 30 degrees C for leatherbacks. Much research remains to be done
in this area of sea turtle ecology: Are pivotal temperatures consistent among
nesting beaches in different parts of the world? Do females "choose"
nesting sites to optimize sex ratios? Does the conservation practice of moving
eggs to hatcheries and beach
zones safe from erosion and predators alter natural sex ratios? What are the
"natural" population sex ratios for hatchlings, juveniles, adults?
The
likelihood of answering these questions is slim until a way can be found to
sex hatchlings without sacrificing them. To complicate matters, juvenile
leatherbacks mysteriously disappear during their developmental years, and the
adults are not seen anywhere consistently except on the nesting beaches.
Although it is certain that nesting turtles are females, this does little to
address the question of what proportion of the population at large is female,
and what proportion is male.
Males are as
enigmatic as juveniles. In some areas such as Pacific Mexico, males are
regularly observed among females in the waters adjacent to the nesting beach.
In other parts of the world such as the insular
These and other questions
highlight the need for expanded pelagic research. A new generation of remote
sensing equipment, more compact and affordable than ever before, promises to
shed light on pelagic behavior. Satellite transmitters and
microprocessor-controlled depth recorders are the wave of the future. We
recently deployed several remote timedepth-recorders (TDRs), developed for use
on marine mammals by Dr. Gerald Kooyman of Scripps Institution of Oceanography,
in an effort to gather information on the diving behavior of leatherback
turtles off their nesting beach at Sandy Point, St. Croix. The turtles turned
out to be remarkable divers, averaging three dives per hour, both day and
night. Submergence time per dive averaged 12 minutes, considerably shorter than
what would have been predicted from previous studies with the smaller chelonid
sea turtles which averaged about 30 minutes per dive. However, maximum dive
depths were far beyond anything we could have imagined averaging 200 feet, but
some exceeded 3300 feet. Equally interesting was the difference in day and
night diving patterns. Day dives were typically deeper, longer and less
frequent than night dives. Turtles spent more time on the surface during the
day, and dive depths were much more variable during the day.
It is not clear why leatherbacks make frequent and robust dives, but we believe they may be foraging. In the tropics, jellyfish are scarce at the surface, but siphonophores and other cnidarians are often associated with a zone of deep-sea biological life called the Deep Scattering Layer (DSL). The DSL is actually a series of zones where zooplankton are concentrated and which reflect and scatter sonic signals. These zooplankters make a rapid vertical migration to the surface at dusk and return to depth (usually below 2000 feet) at dawn. Since the turtles dive more shallowly and more consistently during the night than during the day, they may be foraging within the risen DSL. It is also possible that feeding is not the sole impetus for the diving. Since leatherbacks spend most of their lives in cooler temperate waters, deep diving may serve to lower body temperatures while in warm tropical latitudes. In addition, the really exceptional depths may have been precipitated by predator encounters.
Information
about behavior on a larger scale, such as the post-reproductive movement of
females leaving the nesting beach, is scant. One adult female traveled some 600
miles north to
Scientists
and wildlife managers are increasingly
frustrated by these large gaps in our knowledge about migratory patterns and
life history characteristics. Today there are very few places left where
leatherbacks nest in any numbers, notably Pacific Mexico,
The
former ruling translates into a commitment by the
Fortunately,
international trade, so devastating to hawksbill and green sea turtles, has
never been a serious threat to the leatherback. Thick yellow oil, rendered
from the greasy carapace, is touted as an aphrodisiac in some parts of the
across
international borders in Latin America and
In the
In
most parts of the world the leatherbacks are not killed directly, but indirectly.
Nesting habitat is destroyed by coastal development and other "improvements,"
including jetties, harbor dredging, and heavy recreational use. This global
trend affects not only sea turtles, but also sea birds and nearshore benthic
systems (coastal marshes, seagrass beds, mangrove forests, coral reefs). Nor is
the open ocean spared. At-sea dumping of waste is an enormous and growing problem.
Leatherbacks are stranding from
These are
not remote and distant problems. The thoughtful observer will conclude that
the leatherback sea turtle is threatened by the same forces with which we are
threatening ourselves: poisoned oceans, careless development, an abundance of
garbage. Global extinctions are on the rise. Some say it is a necessary price
to pay. Others believe that man's extinction is inevitable when the oceans and
forests, and the creatures that live there, are gone. Sea turtles and men are
linked in a continuous progression of life. As sea turtles decline, we cannot
help but lose a part of ourselves. And, as the American naturalist William
Beebe has said, "When the last individual of a race of living things
breathes no more, another heaven and another earth must pass away before such a
one can be again."
INTRODUCTION
Sea turtles are highly migratory and ply
the world's oceans. Under the Endangered
Species Act, all marine turtles are listed either as endangered or
threatened . The NMFS has authority to
protect and conserve marine turtles in
the seas, and the U.S. Fish and Wildlife Service maintains authority while turtles are on land.
The Kemp's ridley, hawksbill, and
leatherback turtles are listed as endangered
throughout their ranges. The loggerhead and olive ridley turtles are
listed as threatened throughout their
SPECIES AND STATUS
The Pacific species are loggerhead, green,
leatherback, hawksbill, and olive ridley
turtles. All are also found in the Atlantic Ocean, but the olive ridley
does not commonly enter
Historical data on sea turtle numbers are
limited. In addition, the length of time
that data have been collected has been short when compared with the long life and low reproductive rate of all turtle
species. It is difficult to assess the
long-term status of sea turtles due to the limited data.
The estimated number of female
loggerheads nesting annually in the
southeastern
Historically, the green sea turtle has
supported large fisheries along the
Since 1973, Hawaiian surveys of green
turtles indicate that the estimated
number of turtles nesting annually is about 450-475, and that it is
gradually increasing. No accurate
historical record of this green turtle population exists. The Hawaiian hawksbill turtle population is
very small; only 12-15 nests are recorded
each year. In
ISSUES
Bycatch and Multispecies Interactions
In the North Pacific there were concerns
about sea turtle deaths in the recently
concluded high-seas driftnet fisheries. Turtle bycatch rates were monitored on driftnet vessels by
Turtles are also killed incidentally in
various commercial fisheries. Turtles
are caught and killed in pelagic longline fisheries targeting tunas and billfishes. Conservatively, as many as
11,000 sea turtles may have been killed
annually in offshore shrimp trawls. Turtle mortality from inshore shrimp trawling was not estimated. Fortunately,
turtle excluder devices (TED's) have been
developed for shrimp and fish trawls. TED's enable turtles to escape the
trawl net and prevent them from
drowning. These devices reduce the turtle kill by shrimp trawls by 97% and studies indicate that the use of
TED's minimally reduces shrimp catches.
TED use is presently mandated for most of the Atlantic and
Habitat Concerns
Coastal development is reducing
nesting, egg incubation, and foraging
habitats. Floating tar balls and plastics, if eaten, can harm or kill
sea turtles. The magnitude of these
problems is not fully known, but they occur worldwide, and international cooperation for marine turtle protection
and recovery is required.
Progress
Pacific: The recovery of endangered and
threatened marine turtle populations in
the North Pacific will be enhanced by the United Nations-sponsored moratorium
on large-scale driftnet fishing that went
into effect on 1 January 1993. During 1990 and
1991, when the high-seas driftnet fisheries were carefully monitored by
NMFS and cooperating foreign fishery
agencies, data were collected that have led to a better understanding of the pelagic distribution and
ecology of sea turtles in the North
Pacific.
In the
Significant progress is being made in the
monitoring of Hawaiian green turtles. In
1992, a 5-year series of saturation surveys was completed at
A major factor affecting the recovery of
turtle populations is the mitigation of
commercial fishing/sea turtle interactions. The incidental capture of sea
turtles in various commercial fisheries
has been studied and summarized and was the focal point of a meeting at the recent 13th Annual Sea
Turtle Symposium. Recent legislation has
allowed NMFS to use observers in selected fisheries to document the occurrence
of incidental turtle captures. Also,
several new TED models have been recently tested and approved for commercial use, and research
continues on the development of a new
TED design which would accommodate small inshore turtles.
Considerable progress has been made
concerning inshore juvenile developmental
habitat research and remote sensing. NMFS research projects have been
started on juvenile ridleys and greens
in the Cedar Keys and