A Solitary Flagellate: Euglena Phylum Sarcomastigophora
Subphylum Mastigophora
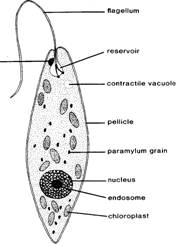
Euglena (figure
5.1) is a common green flagellate often found in the greenish surface scum of
standing or slowly moving water. Euglena
is an enigmatic organism with a curious mixture of plant and animal
characteristics, and, therefore, sometimes is considered to represent a borderline
case between the plant and animal kingdoms. Euglena
is smaller than Amoeba and Paramecium and, therefore, the details
of its internal structure are more difficult to observe.
·
Prepare a wet mount from a
culture of living Euglena and observe
the locomotion of an active specimen under your compound microscope. The active
swimming movements result from the beating of the long flagellum, which pushes
the organism through the water. A second, shorter flagellum is present within
the flagellar pocket, but does not aid in the swimming movements. At certain
times Euglena also exhibits another
type of wormlike locomotion during which waves of contraction pass along the
body in a characteristic fashion. This type of locomotion is peculiar to Euglena and related organisms, and is
appropriately termed euglenoid movement
or metaboly. It appears to
result in part from the elasticity of the thick outer covering of the body, the
pellicle.
After the wet mount begins to dry out, temporarily
immobilizing some of your specimens, study the anatomy of a stationary Euglena. You will also find it useful to
supplement your observations with the study of a prepared microscope slide.
Identify the following structures under high power on your
compound microscope: (1) pellicle, the
thick outer covering of the body; (2) chloroplasts
with green chlorophyll; (3) nucleus,
exhibiting a large central endosome in
stained preparations; (4) flagellar
pocket; (5) contractile vacuole; (6)
a red stigma, or eye spot; (7) the
long anterior flagellum; and (8) paramylum grains, a type of starch that
represents stored food materials.
Euglena is quite sensitive to light, and changing the light intensity
tends to cause the Euglena to move
away. Bright light tends to make this protozoon remain stationary.
·
After you have completed
your observations of the living specimen, add a drop of Lugol's solution
(iodine and potassium iodide). This solution will kill the specimen and stain
the flagellum to make it more readily visible.
As suggested by the presence of chloroplasts, the nutrition of Euglena is normally autotrophic;
organic molecules (sugars) are synthesized from inorganic nutrients absorbed
from the medium. Light from the sun provides the energy necessary for this
process.
Biochemical tests have shown the paramylum granules to be a
form of starch similar to that found in plants. Thus, both the presence of the
chloroplasts and the storage of a plantlike form of starch indicate a close
relationship of Euglena and its
relatives to the plant kingdom.
·
Some species of Euglena are also able to survive, grow,
and reproduce in the dark with no visible evidence of chloroplasts, chlorophyll,
or stored food materials.
How
might such organisms obtain their food?
A Colonial (?) Flagellate: Volvox
Phylum
Sarcomastigophora
Subphylum Mastigophora
Volvox (figure
5.2) is a common green alga that often occurs in great numbers in freshwater
ponds and lakes. Although Volvox is
photosynthetic and is sometimes considered to be an alga, it is often studied
in zoology courses because it illustrates the organization of a simple colonial
(or multicellular) organism well and because of its usefulness in illustrating
one popular theory for the evolution of multicellular organisms from
unicellular ancestors. It also demonstrates some basic similarities between
plants and animals.
The spherical green Volvox
are large enough to be seen swimming near the surface of a pond or in a
laboratory culture even without a microscope.
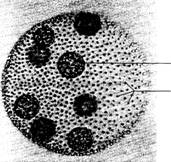
Fig. 5.2
Volvox,
asexual spheroid. (Courtesy of Carolina Biological Supply Company,
\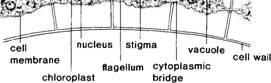
Fig. 5.3
Volvox
cell structures, longitudinal section of cells at surface of spheroid.
Obtain
some living Volvox from a culture and
prepare a wet mount. Be sure to add a few bits of broken coverslip or sand
grains to protect the spherical Volvox bodies
from being crushed by the weight of the coverslip. Observe the spheroid shape
of Volvox, its swimming movements,
and its prominent green color.
The spherical Volvox bodies are usually called colonies in
textbooks, but recent studies have suggested that they are more similar to
multicellular individuals. The coordination of cells and cellular function
within the spheroid body is much greater than in most other colonial algae and
protozoons; thus, recent workers use the term spheroids for the Volvox body.
The hollow Volvox spheroids
average about 0.5mm in diameter and have many small green cells embedded in
their outer walls (see figure 5.3). Each of the tiny body cells of Volvox contains a nucleus, a contractile
vacuole, a green chloroplast, and
two whiplike flagella. Some or all
of the cells (depending on the species) may also have a red stigma (light-sensitive spot). The
flagella project outward from the surface, and their beating keeps the spheroids
in a constant spinning motion. Although the somatic cells of Volvox are very small and difficult to
observe except with special microscopic preparations, the adjacent cells in
some species of Volvox are connected
by thin cytoplasmic bridges or
strands. Other species of Volvox, however,
lack these intercellular connections.
·
Add a drop of 0.1 %
methylene blue solution to a wet mount of Volvox
and study under high power on your compound microscope. Can you observe any
cytoplasmic bridges?
The green color of the spheroid individuals results from the
presence of a chloroplast in each cell. What can you therefore infer about the
nutrition of Volvox?
Within the spheroid you should be able to observe one or more
large reproductive cells or gonidia. Reproduction
in Volvox involves both sexual and asexual processes. In asexual development,
embryos are formed from the gonidia.. Locate several gonidia within the
interior of an asexual parent individual (see figure 5.4).
During asexual development, the gonidia undergo a series of
cell divisions remarkably like those seen in the embryonic development of many
animal species. See if you can locate several different developmental stages of
gonidia in the living specimens provided.
The
beginning of sexual reproduction can be recognized when the gonidia form male
and/or female spheroids. Most species of Volvox
have separate male and female individuals (i.e., are dioecious, "two houses"), but some species produce both
eggs and sperm in the same individual and are thus monoecious ("one house"). Figure 5.4 also shows both male
and female individuals.
The living cultures available for
study in the laboratory are usually all asexual. You should study the demonstration
chart to learn about the life cycle of Volvox.

Fig. 5.4
Volvox,
male, female, and asexual spheroids. (Photograph by Barbara Grimes.)
Study the structure of Volvox
using the living specimens provided in the laboratory. In a drop of water
on a clean microscope slide, observe the swimming of Volvox spheroids first
under low power, and then add a coverslip over the water drop and observe more
details of the structure of the colony under higher magnification. Observe
somatic cells, gonidia, eggs, sperm, and zygotes, using both the living
specimens and the demonstration materials as necessary.
Evolution of Multicellularity
Volvox and
several related green flagellates are often studied as models to illustrate one
popular theory for the evolution of multicellular organisms. Most scientists believe
that multicellular organisms arose from some unicellular form. The particular
kind of unicellular organism is not known because this major evolutionary step
took place more than 600 million years ago in the Precambrian Era. No
well-preserved fossils have been found that actually document this transition
from one to many cells, so biologists have searched among living plants and
animals to seek models that might
help their understanding of early evolution.
Volvox and several related green algae comprise the most popular
model discussed by scientists. These related forms exhibit a graded series of
solitary and colonial forms of increasing complexity and are called the Volvocine Series (figure 5.5). Among
the important genera of algae comprising the series are: Chlamydomonas, Gonium, Pandorina, Eudorina, Pleodorina, and Volvox.
Other Mastigophora
Other
important mastigophorans include the dinoflagellates, the symbiotic
flagellates that inhabit the digestive tracts of termites and the wood roaches,
some peculiar flagellates that may be related to sponges, and several important
parasites of humans.
Dinoflagellates are found in both fresh and marine waters; many species form a
characteristic outer covering, called a test,
that is made of cellulose. Certain freshwater dinoflagellates may cause an
unpleasant odor or taste in human water supplies. Gonyaulax and Gymnodinium are
two marine dinoflagellates often associated with the red tides of coastal
waters of North America, Europe, and
Fig. 5.6 Symbiotic flagellates from termite gut. (Courtesy of
Carolina Biological Supply Company,
Some flagellates live as symbionts in the digestive tracts of
wood roaches and termites (figure 5.6). Experiments have shown that the
termites lack the digestive enzymes necessary to digest the cellulose in the
wood they eat. The flagellates ingest splinters of wood and form food vacuoles
around them. Later, digested products from the breakdown of the cellulose are
released from the protozoa and provide nutrients for the termites. Termites
from which the flagellates are experimentally removed soon die of starvation no
matter how much wood they ingest. The flagellates benefit from the continuous
supply of cellulose and from the suitable anaerobic environment of the host
hindgut. Such a mutually beneficial symbiotic relationship is called mutualism.
Proterospongia is a colonial flagellate with species that closely resemble the
flagellated collar cells, or choanocytes, characteristic of sponges (see
chapter 6). Some biologists have suggested that the sponges may have evolved
from some ancient protozoon similar to Protero
spongia.
Still other mastigophorans are parasites. Trypanosoma and
Leishmania are two important genera
that include several serious human parasites. Trypanosoma has a thin, undulating membrane connecting its long,
whiplike flagellum with its body (see figures 5.7 and 5.8). Several species
of Trypanosoma cause sleeping
sickness and other diseases in humans. Leishmania
includes species that cause severe diseases in tropical areas of
An Amoeba: Amoeba proteus
Phylum Sarcomastigophora
Subphylum Sarcodina
Amoeba proteus (figure 5.9) is a protozoon found in ponds and streams. It
often occurs on the undersides of plant leaves, and among diatoms and desmids.
The transparent amoeba constantly changes shape by extending pseudopodia,
footlike extensions of the cytoplasm, which serve for locomotion and in food
capture. Amoeba proteus feeds on
bacteria, small algae, and small protozoons.
In feeding, an advancing pseudopodium flows over one or more
food organisms to trap the food in a waterfilled cup. The opening of the food
cup then narrows until the food is completely enclosed in a food vacuole.
Demonstrations
1. Large flagella in other Mastigophora, such as Peranema.
2.
Microscope slide of trypanosomes.
3.
Microscope slide of symbiotic flagellates from digestive tract of termite or
wood roach.
4.
Microscope slide of dinoflagellates.
5. Microscope slide showing cell walls in Volvox.
6. Chart
illustrating the life cycle of Volvox.
7.
Microscope slides with related volvocine flagellates,
such as: Gonium,
Pandorina, Eudorina, and Pleodorina.
white blood cell
r
Fig. 5.7 Trypanosoma, blood smear. (Courtesy of Carolina
Biological Supply Company,

Fig. 5.8 Trypanosoma. Magnification 3,525X. (Scanning electron
micrograph by Louis de Vos.)
I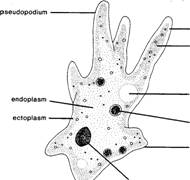
Fig. 5.9 Amoeba proteus.
Prepare a wet mount to
study living amoebae under your compound microscope.
Preparing a Wet Mount and a Hanging Drop. Obtain a clean microscope slide and add a drop of amoeba
culture solution to the center of the slide. Take care to withdraw the drop of
culture solution from the bottom of the culture dish or jar with a clean
eyedropper or pipette. The amoebae are slightly heavier than the culture
solution and are usually concentrated on the bottom of the vessel. Add a few bits of broken coverslip or grains
of sand around the periphery of the drop to protect your specimens from being
crushed, then carefully cover your preparation with a coverslip. This
preparation is called a wet mount.
Another method of studying living Protozoa is to prepare a hanging drop. With this method you
place a drop of the amoeba culture in a coverslip and invert the coverslip
over the cavity of a depression slide. Both types of preparations can be
observed for long periods of time if the outside edge of the coverslip is
coated with petroleum jelly before it is placed in position on the microscope
slide. Be sparing in your use of the petroleum jelly; avoid getting it into
the culture droplet and on your microscope lens.
Study your preparation under the low power of the compound
microscope to observe the general appearance of the amoeba. For best
observation of a living amoeba, reduce the illumination to a minimum since
living specimens are nearly transparent and almost invisible in bright light.
Search your slide carefully for a specimen before discarding it or asking your
instructor for a new preparation.
Locate an actively moving amoeba and note its constantly
changing shape. The long, fingerlike projections are pseudopodia ("false feet"). Observe the lack of permanent
orientation of the body of an amoeba; any portion may temporarily be anterior,
posterior, right, or left.
With the
aid of figure 5.9, identify and study the following structures found in the amoeba.
1. Endoplasm the
inner granular region, which forms the bulk of the cytoplasm.
2. Ectoplasm the
thin layer of clear cytoplasm which surrounds the endoplasm.
3. Cell membrane the
outer membrane surrounding the amoeba. Sometimes also called the plasmalemma.
4. Plasmagel the
stiff, jellylike, granular outer layer of colloidal endoplasm in the gel state.
5. Plasmasol the
central mass of colloidal endoplasm in a fluid, or sol state. Note the streaming movements within the plasmasol.
6. Nucleus a
transparent structure with no fixed position in the cell. It has the shape of
a biconcave disc and often exhibits a folded or wrinkled appearance. Examine
also the nucleus in a stained microscope slide of Amoeba proteus. Observe the darkly staining granular chromatin
material within the nucleus.
7. Contractile vacuole a clear vacuole
found in the endoplasm which collects excess water from the surrounding
cytoplasm and body. Shortly after one contractile vacuole discharges its
contents at the cell surface, a new contractile vacuole forms. Where are the
new contractile vacuoles formed? Formerly, it was believed that the contractile
vacuole also played an important role in the excretion of waste products from
protein metabolism, but recent evidence has not supported this belief, and
most specialists now agree that the contractile vacuole functions primarily in
maintaining water balance in the cell (osmoregulation)discharges it outside the
cell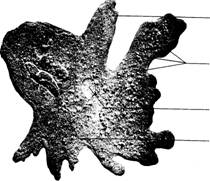 Fig. 5.10 Pelomyxa carolinensis. A
large multinucleate amoeba. (Courtesy of Carolina Biological Supply Company,
Fig. 5.10 Pelomyxa carolinensis. A
large multinucleate amoeba. (Courtesy of Carolina Biological Supply Company,
8. Food vacuoles vacuoles containing bits
of ingested food and the digestive enzymes that act to break down these food
materials into soluble materials that can be utilized by the amoeba. How are
the food vacuoles formed? How are the undigested contents of a food vacuole
disposed of after digestion has taken place?
Amoeboid Movement. Amoeba moves about by extending pseudopodia into which some of the
innermost cell contents flow. Various kinds of amoebae form pseudopodia of
different size and form. These pseudopodia are important in feeding, support,
and locomotion. The mechanism of amoeboid movement has been studied by many
scientists because of its intriguing nature and because similar movements
occur in many other kinds of cells, including human leucocytes. Also,
scientists now believe that amoeboid movement may be closely related to the
phenomenon of cytoplasmic streaming, which occurs in virtually all kinds of
living cells.
The movement of an amoeba is accomplished by the forward flow
of the relatively liquid plasmasol from
the center of the amoeba toward and into an expanding pseudopodium. Around the
periphery of the pseudopodium, the
plasmasol
changes into a stiff plasmagel. Thus,
the plasmasol moves the pseudopodium forward, and the plasmagel serves to fix
it in position.
Recent biochemical and biophysical studies have demonstrated
that the mechanism of amoeboid movement is similar to that in muscle
contraction. Contractile proteins similar
to the actin and myosin found in vertebrate muscles are present in the
cytoplasm of an amoeba. Amoeboid movement results from folding, unfolding,
polymerization, and depolymerization of these proteins.
∎ Study the locomotion of an Amoeba on your microscope slide and also the pattern of its
internal protoplasmic movements. In an active specimen, locate and carefully
follow the movement of some granules in the plasmagel at the temporary
posterior end. Observe how the plasmagel of the endoplasm changes into
plasmasol, which flows forward and then changes into the gel state again, just
back of the tip of the forming pseudopodium.
Reproduction. The
reproduction of Amoeba proteus occurs
only through the asexual process of binary fission. The nucleus and cytoplasm
of a parent cell divide to form two daughter cells approximately equal in size.
Thus, each of the daughter cells is genetically identical to the parent
cell-excluding the rare occurrence of a mutation in one of the daughter cells.
Other Sarcodina
Many
members of this group of Protozoa are more specialized than Amoeba. Pelomyxa carolinensis is a large
multinucleate amoeba often studied in zoology classes (figure 5.10). Numerous
species of amoebae live in shellsaperture
Fig. 5.11 Test of a freshwater amoeba. (Scanning electron micrograph by F. W.
Harrison.)
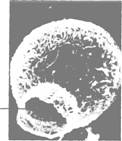

Fig. 5.12 Actinosphagerium.
(Courtesy of Carolina Biological Supply
Company,
or tests
which they secrete, or which they form from sand grains or other materials
(figure 5.11). Difflugia and Arcella are two common testate amoebae
found in freshwater ponds and streams. Other species of amoebae are parasites
or symbionts in the digestive tracts of various animals. Entamoeba histolytica, an important parasite of humans, is the cause
of amoebic dysentery, a disease often spread by drinking water or by eating raw
vegetables contaminated by human wastes in parts of the world with poor
sanitary facilities.
Some freshwater members of the Subphylum Sarcodina have many
long, thin pseudopodia supported by axial rods of microtubules. Actinosphaerium (figure 5.12)
is a
common freshwater example of a group called heliozoans because of the
resemblance to the sun and its rays of sunlight.
Members of three marine classes of this subphylum, called radiolarians, form skeletons or tests
of silicon and/ or strontium compounds and exhibit many beautiful shapes
(figure 5.13). The radiolarians are among the oldest known protozoa, and their
tests are abundant in marine sediments in many parts of the world.
The foraminiferans (figure 5.14), representing another class of
sarcodines, are an ancient and important group of marine sarcodines which form
tests of calcium carbonate or other materials. The shells of foraminiferans


![]() Fig. 5.13 Radiolarian tests.
(Courtesy of Carolina Biological Supply Company,
Fig. 5.13 Radiolarian tests.
(Courtesy of Carolina Biological Supply Company,
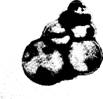 Fig. 5.14 Foraminiferan tests.
(Photograph by Barbara Grimes.)
Fig. 5.14 Foraminiferan tests.
(Photograph by Barbara Grimes.)
accumulate on the sea bottom and
contribute to the formation of chalk and limestone.
∎ Observe several of
these other types of Sarcodina among the demonstrations.
Demonstrations
1. Models and charts of
Amoeba proteus.
2. Culture of Amoeba under a stereomicroscope.
3.
Microscopic slides with testate and parasitic representatives of the Subphylum Sarcodina,
such as Arcella, Difflugia, Entamoeba
histolytica, Actinosphaerium (a heliozoan), and Globigerina (a foraminiferan).
A Ciliate: Paramecium caudatum Phylum
Ciliophora (Ciliata)
Paramecium (figure
5.15) is a large, common, ciliated protozoon often found in water containing
bacteria and decaying organic matter. There are several species of Paramecium that differ in various
details of structure and that range in length from about 120-300 microns. The
laboratory directions provided here are based upon Paramecium caudatum, a species frequently used for laboratory
study and experimentation, but they will also apply, with minor modification
(such as body size and number of micronuclei), to the study of other species
of Paramecium.
∎ Obtain a drop of Paramecium
culture in a clean pipette and place it on a clean microscope slide with a
similar-sized drop of methyl cellulose solution (or other similar agent) to
slow movement. Methyl cellulose is a viscous material and serves mechanically
 I
I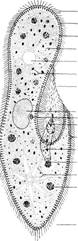 Fig. 5.15 Paramecium caudatum.
Fig. 5.15 Paramecium caudatum.
Fig. 5.16 Paramecium, nigrosin
stain to illustrate sculpturing of bilayered pel icle. (Photograph by Barbara
Grimes.)
to slow the swimming of the fast-moving Paramecium. Add a coverslip and observe
your preparation under low power with your compound microscope. Note the form,
color, and behavior of the animals in your preparation. Observe the
slipper-shaped body with an oral groove beginning
at the anterior end and running diagonally across the anterior portion of the
animal. At the posterior end of this groove is the cytostome, or "cell mouth," through which food particles
are passed as a result of the action of the specialized oral cilia lining the
oral groove.
Paramecium
is much more complex in its structure than Amoeba. Select a large, immobile, or
slowly moving specimen, and with the aid of figure 5.15, identify and study the
following structures.
1. Cilia the numerous cylindrical protoplasmic extensions
that cover the surface of the Paramecium and
that function in locomotion and in food gathering.
2. Pellicle the
thick outer covering of the body through which the cilia project. The pellicle
has a complex structure, but its details are difficult to observe without
special techniques. Figure 5.16 shows some of the surface depressions in the
bilayered pellicle.
3. Trichocysts tiny,
rodlike structures embedded in the cortical (outer) cytoplasm beneath the
pellicle. When properly stimulated, the trichocysts discharge their contents
and form long threads. There is some evidence that the trichocysts may serve as
a defense against predators, and they also serve to anchor the animal during
feeding. In other types of ciliated protozoa, trichocysts have been found to
have still other functions. Observe the microscopic demonstration of
discharged trichocysts.
4. Macronucleus the
large nucleus located near the center of the cell. Since it is transparent in a
living animal, the structure of the macronucleus is best studied in a stained
preparation. Experiments have demonstrated that the macronucleus controls most
metabolic functions of the cell.
5. Micronucleus a
smaller nucleus located close to and lying partly within a depression on the
oral side of the macronucleus. The micronucleus is involved primarily in the
reproductive and hereditary functions of the animal. This presence of two
distinct types of nuclei is called nuclear
dimorphism and is a condition found only in the Phylum Ciliophora. Paramecium caudatum has only a single
micronucleus, but some other species of Paramecium
have two or more micronuclei. As with the macronucleus, the structure of
the micronucleus is best studied in a prepared microscope slide.
6. Contractile vacuoles
two clear, slowly pulsating vesicles located near each end of the body.
Each contractile vacuole is surrounded by several radiating canals (not often seen in ordinary student preparations)
which collect water from the surrounding cytoplasm. Observe the behavior of
the contractile vacuoles. Are they fixed in position? Do they contract
alternately or simultaneously? The function of the contractile vacuoles in Paramecium is the same as in Amoeba, the collection and discharge of
excess water from the cell. Freshwater protozoa often have contractile
vacuoles; marine protozoa generally lack them. How would you explain this
difference?
7. Cytostome (cell
mouth) a permanent opening near the posterior end of the oral groove through
which food is passed.
8. Cytopharynx a
short tube extending from the cytostome posteriorly and downward into the cytoplasm
where food vacuoles are formed.
9. Food vacuoles vacuoles
located within the cytoplasm where they are carried by the streaming movements
of the cytoplasm. Undigested materials are discharged through the cytopyge, or
anal pore, located posterior to the oral groove.
Feeding
Paramecium is
a filter-feeding organism and normally feeds on bacteria and yeast cells
collected by a specialized food-collecting apparatus. An oral groove extends diagonally back along the body to a
funnel-shaped cytopharynx. Food is
swept along the oral groove by the action of specialized cilia lining the
groove, is passed through the circular cytostome
("cell mouth") at the opening of the cytopharynx, and is passed
through the cytopharynx into a newly forming food vacuole.
∎ Prepare a wet mount with a drop of Paramecium culture to study the feeding
process. Add a small amount of congo red stained yeast with the tip of a
toothpick or clean dissecting needle. Try to pick up the smallest amount of
yeast possible on the toothpick; too much yeast will cloud your preparation and
obscure the Paramecium.
With this preparation, you can study the movement of the food
particles, the formation of food vacuoles, and the subsequent movement of the
food vacuoles within the cytoplasm. After the food vacuoles are formed,
digestive enzymes are released into them and chemical digestion of the food
particles begins. Note the color change in the vacuoles as the enzymes work.
The color change is due to a change of pH in the vacuoles. Where do the digestive
enzymes come from? Why don't they digest the other materials in the cell such
as mitochondria and ribosomes? The diffusible products of digestion are
released into the cytoplasm, and the undigestible remains are discharged at a
specific site on the surface of the animal. This site is the cytopyge, or cell anus.
Cilia and Flagella
Most of the surface of Paramecium
is covered by thin, hairlike projections called cilia (singular: cilium).
Cilia are extensions of the cortical (outer) cytoplasm of the cell and play
important roles in feeding and locomotion.
A great deal has been learned in recent years about the
structure and function of cilia. These studies have revealed that cilia are
closely related to the flagella (singular: flagellum) found on the surface of
other kinds of protozoa. The structural differences between cilia and flagella
are minor. When the projections are short and numerous, they are called cilia.
When they are long and few, they are flagella. Cilia generally exhibit a
relatively simple back and forth movement. The movements of flagella are often
more complex and may involve a series of helical waves propagated along the
flagellum.
Both cilia and flagella have a common basic structure. A
cross section reveals an outer membrane enclosing a circle of nine pairs of microtubules and two single
microtubules in the center of the cilium or flagellum. This basic pattern
is found in all cilia and flagella, not only among the protozoa but also on the
gills of molluscs, the ciliated epithelium lining the trachea of vertebrates,
and the tail of spermatozoa.
Recent biochemical studies have also demonstrated that the
movements of cilia and flagella involve contractile
proteins similar to those found in striated muscle. This is another
important illustration of the basic similarity of all living organisms.
Reproduction
Paramecium reproduces by a simple type
of asexual reproduction in which the parent divides into two equal daughter
cells. This type of asexual reproduction is termed transverse fission and is found in many kinds of protozoa. Living
specimens are occasionally seen in the process of fission, but the details of
fission are best studied in a stained microscope slide. Obtain a prepared slide
of Paramecium in fission and observe
the nuclei. During fis sion, the micronucleus first divides by mitosis, and the macronucleus later
divides by amitosis. No visible chro
mosomes are formed in the macronucleus; the macronu cleus simply constricts,
and the two portions separate. Macronuclear division is followed by cytoplasmic
division (cytokinesis). The process of fission may be completed rapidly, and
under optimal conditions, Paramecium can
reproduce asexually two or more times per day.
∎ Draw the representative stages of fission in Paramecium as indicated on figure 5.17.
Unlike Amoeba,
Paramecium can also reproduce sexually. The specialized type of sexual
process exhibited by Paramecium is
called conjugation (figure 5.18). During this process, two individuals come
together, adhere by their oral surfaces, undergo a complex series of changes in
both the macronuclei and the micronuclei, exchange a single pair of micronuclei
(one from each cell), separate, and resume asexual reproduction. Following the
exchange of micronuclei in each Paramecium,
the newly introduced micronucleus fuses with another (nonmigrating) micronucleus.
Thus, there is an exchange of hereditary material and a subsequent fusion of
hereditary material from the two parents, a situation analogous to that of
ordinary sexual reproduction studied earlier.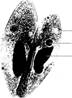 Fig. 5.18 Paramecium in conjugation.
(Courtesy of Carolina Biological Supply Company,
Fig. 5.18 Paramecium in conjugation.
(Courtesy of Carolina Biological Supply Company,
∎ Examine a prepared slide or a demonstration of Paramecium in conjugation.
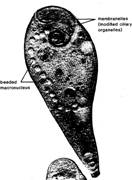

Fig. 5.20 Didinium, a carnivorous
ciliate ingesting a Paramecium. (Courtesy
of Carolina Biological Supply Company,
Fig. 5.19 Stentor.
(Courtesy of Carolina Biological Supply
Company,
Other Ciliata
The
Phylum Ciliophora (Ciliata) is a large and diverse group of protozoa. One
important form is Stentor (figure
5.19), a large, trumpet-shaped ciliate which is common in lakes, ponds, and
streams, and which has been used in many experimental studies. Stentor has a spiral array of complex
ciliary organelles leading to its cytostome and a beaded macronucleus. Didinium (figure 5.20) is a barrelshaped
predaceous ciliate with a voracious appetite. It feeds on other ciliates
including Paramecium. A hungry Didinium can eat a Paramecium every two hours.
Spirostomum (figure 5.21) is a long, wormlike ciliate that has
contractile fibrils that seem to function in a way similar to striated muscle
fibrils. Tetrahymena (figure 5.22) is
a small, ovoid ciliate that has been used in many experimental studies of
biochemistry and genetics.
Vorticella (figure 5.23) is a sessile form with a long, contractile stalk
which attaches to submerged stones, shells, plants, animals, and other objects.
Some relatives of Vorticella, such as
Carchesium and Zoothamnion, form stalked colonies.
Podophyra
is a suctorian, a specialized group
of sessile ciliates that have suctorial
tentacles in their mature stages and are predators of other ciliates. Cilia
are found only on juvenile stages of the suctorians.
1.
Models and charts of Paramecium.
2. Stained slide showing pellicle of Paramecium.
3. Stained slide to show discharged trichocysts.
4.
Stained slides with representative members of the
Class Ciliata, such as Stentor, Euplotes, Tetrahymena, Vorticella,
Didinium, Blepharisma, Trichodina, and Podophyra.
Phylum Apicomplexa
Members of this group were formerly included among the
Sporozoa, but recent investigations have indicated that they should be
considered a separate phylum. All members of this phylum are parasitic on
other organisms.
Many species in this group parasitize invertebrate animals
such as earthworms, crabs, and oysters, but the most important species are
parasites of vertebrates, including humans. Among the most important
sporozoans are Plasmodium and Eimeria. Several species of Plasmodium cause various forms of malaria in humans and other animals.
Demonstrations
Chapter 5

Fig.
5.21 Spirostomum, a ciliate with contractile fibers and a flexible pellicle that exhibits
a peculiar, wormlike locomotion. (Courtesy of Carolina Biological Supply
Company,
Eimeria is a genus of sporozoan parasites that causes coccidiosis in birds, rabbits, and
other animals. This dis cytostome ease
has great economic impact on the poultry industry. Eimeria, like Plasmodium, has
a complex life cycle that includes both sexual and asexual forms. Study the demonstrations
illustrating the life history and importance of Eimeria and coccidiosis.
The Malaria Parasite: Plasmodium
More
than 50 species of Plasmodium have
been described. All are parasites of vertebrate animals, including amphibians,
reptiles, birds, and mammals. Four species cause human malaria, P. vivax, P. ovale, P. malariae, and P. falciparum. The life cycles of these
species are all similar.
Malaria, one of the most serious and debilitating of human
diseases, has had an important role in history from the fall of the
Protozoa
Fig. 5.23 Vorticella. A sessile ciliate with
along contractile stalk for attachment and a ring of cilia surrounding the
mouth. (Courtesy of Carolina Biological Supply Company,
Life Cycle of Plasmodium
The life cycle of Plasmodium
(see figure 5.24) is complex like those of other apicomplexa and includes
several generations with both sexual and asexual reproduction. The life cycle
can best be understood by starting with the zygote in the gut of a mosquito,
one of the two hosts necessary for the completion of the life cycle.
The zygote becomes motile and passes through the lining and
wall of the stomach or midgut of the mosquito and is now called an ookinete.
The ookinete then rounds up and encysts on the outside of the gut wall and is
called an oocyst. After several days of growth, the oocyst divides internally
to form several hundred sporozoites. The sporozoites escape by rupturing the
external wall of the oocyst and migrate through the hemocoel to the salivary
glands of the mosquito.
When a mosquito bites a human, the sporozoites and the
mosquito's salivary secretions are injected into this host. Sporozoites that
find their way into the human bloodstream are eventually carried to the liver,
where the sporozoites enter host cells. Inside the host liver cells, the
sporozoites transform into amoeboid multinuclear schizonts and feed upon the
contents of the host liver cells. The schizonts reproduce asexually to form
many merozoites (the next stage in the life cycle), escape from the liver
cells, and, in some cases, invade other liver cells to repeat the process.
Merozoites escaping into the
bloodstream penetrate erythrocytes to initiate the erythrocytic phase of the
life cycle. Parasitic stages in the liver prior to entry into the erythrocytes
are termed the exoerythrocytic phase.
The developing Plasmodium inside the erythrocytes
exhibit a characteristic morphology, as seen in Giemsastained microscope
preparations, and are recognized by their red nucleus and blue ring-shaped
cytoplasm (see figure 5.25). This characteristic morphology is very helpful in
the laboratory diagnosis of malaria. The merozoites in the erythrocytes undergo
further asexual reproduction within the erythrocytes. Later, the merozoites
rupture the wall of the erythrocytes, escape into the blood, and enter most
erythrocytes. This multiplication process may be repeated several times, so
that an enormous number of parasites are produced within the host. The rupture
of the erythrocytes by the merozoites also releases accumulated toxic wastes
from the parasites and causes the symptomatic chills and fever commonly
associated with human malaria.
Some of the merozoites develop
into sexual forms instead of repeating the asexual merogony. These sexual forms
develop within the erythrocytes and become microgametocytes (male) and
macrogametocytes (female) (figure 5.26). These stages are the progenitors of
the male and female gametes, and represent the start of the sexual portion of
the life cycle.
If a mosquito bites an infected
host and ingests infected erythrocytes, the gametocytes pass into the mosquito's
stomach, where they mature into microgametes and
162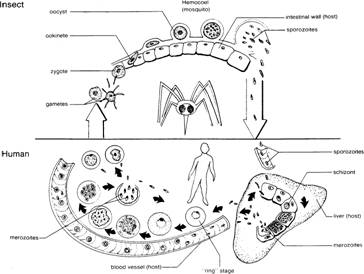
Fig. 5.24 Life cycle of Plasmodium.
macrogametes. The microgametocytes
develop flagella like outgrowths, which break free, become motile, and
fertilize macrogametes. The fertilized macrogametes, or zygotes, then invade
the gut wall of the mosquito to start uninfected red the cycle.
∎ Study the microscope slides with blood smears prepared
with blood from humans infected with Plasmodium
vivax or a similar species. Identify as many stages in the life cycle of Plasmodium as you can from your own
slides and the demonstration slides. Observe the changes in morphology of the
parasite during its development in the human erythrocytes. With the aid of
figure 5.24, try to relate the portion of the Plasmodium life cycle in human erythrocytes to the other parts of
the life cycle completed in the mosquito and in the exoerythrocytic stages in
the human. Why do you think Plasmodium requires
two hosts to complete its life cycle? What special adaptations for life as a
parasite can you identify in Plasmodium?
List several of these adaptations for parasitism in table 5.1. blood cell
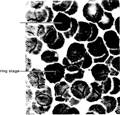
Fig. 5.25 Plasmodium. Ring stage
inside human erythrocyte. (Courtesy of Carolina Biological Supply Company, 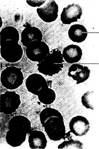
Key Terms
Cilia cylindrical cytoplasmic extensions from the
surface of certain protozoa and of some metazoan cells. Serve in locomotion
and feeding of ciliated protozoa .uninfected Basically similar to flagella, but
shorter and more nu blood cellmerous. All have the universal internal 9 + 2
pattern of microtubules. Singular: cilium.
Coccidiosis disease
of birds, rabbits, and other animals caused by members of the protozoon genus Eimeria.
Macrogametocyte
Conjugation
a specialized type of mating, nuclear exchange,
and nuclear reorganization characteristic of ciliates. A form of sexual
reproduction.
Contractile
vacuole an organelle found in many freshwater
protozoons that serves in osmoregulation (water balance).
Cytostome
the "cell mouth" found in many
protozoa, including ciliates, some flagellates, and some apicomplexa. In
ciliates, the cytostome is often surrounded by specialized ciliary feeding
organelles.
Flagella
cylindrical cytoplasmic extensions from the
surface of certain protozoa and some metazoan cells. Function in locomotion and
feeding of mastigophorans. Similar to cilia but longer and usually fewer per
cell. Singular: flagellum.
Gonidia specialized reproductive cells in Volvox. Singular: gonidium.
Macronucleus
the large metabolic nucleus typical of ciliates.
Often has a characteristic shape. Divides amitotically by pinching in two.
Contains many duplicated sets of genes (polyploid).
Malaria disease
of humans and other animals. Caused by members of the protozoon genus Plasmodium, which invade the blood and
other tissues of the hosts.
Micronucleus the small reproductive nucleus in ciliates. Some ciliates have
more than one micronucleus. Usually divides by ordinary mitosis.
Nuclear dimorphism having two distinct types of nuclei in the same cell.
Characteristic of the ciliates (e.g., with macronucleus and micronucleus).
Pseudopodia the
protoplasmic extensions, "false feet," of the Sarcodina used for
locomotion and in feeding. Various types of sarcodines have pseudopodia specialized
for certain purposes or modes of life. Singular: pseudopodium.
Demonstrations
1. Microscope slides with selected stages in the life cycle Stigma
a light-sensitive spot found in certain flagellated
of Plasmodium. protozoa,
such as Euglena and Volvox. 2. Chart illustrating the life
cycle of Plasmodium.
3.
Microscope slide of Eimeria.
4. Chart illustrating the life
cycle of Eimeria.
Fig. 5.26 Plasmodium. Developing macrogametocyte in human
erythrocyte. (Courtesy of Carolina Biological Supply Company,